
Exhibition
Step into Cognitive Manufacturing
The IMA Ever‑Evolving System invites you into a new era of manufacturing.
IMA for:
Hover an industry on the left menu, see the full list or type your search:
IMA for: Pharmaceutical
IMA for: Food
IMA for: Coffee
IMA for: Tea & Herbs
IMA for: Personal Care
IMA for: Tissue & Nonwoven
IMA for: Tailored Technologies
IMA for: E-Commerce
IMA for: Automation
Technologies
Machines
Lines
IMA: Services
IMA: Corporate
IMA: Contacts
IMA for:
Hover an industry on the left menu, see the full list or type your search:

Exhibition
Step into Cognitive Manufacturing
The IMA Ever‑Evolving System invites you into a new era of manufacturing.
IMA for: Pharmaceutical
IMA for: Food
IMA for: Coffee
IMA for: Tea & Herbs
IMA for: Personal Care
IMA for: Tissue & Nonwoven
IMA for: Tailored Technologies
IMA for: E-Commerce
IMA for: Automation
Technologies
Machines
Lines
IMA: Services
IMA: Corporate
IMA: Contacts
Technology



With over 50 years of experience, IMA designs and manufactures freeze-drying systems tailored to the needs of pharmaceutical and biotech production, from laboratory-scale development to commercial manufacturing.
Based on a modular and customizable architecture, IMA Life’s freeze dryers support a wide range of applications, ensuring shorter lead times, reduced operating costs, and enhanced product quality. The portfolio includes industrial, small-scale and laboratory freeze dryers, also suited for complex formulations.
Freeze-Drying
All systems are fully compliant with GMP Annex 1, ASME BPE and other major international standards, ensuring optimal cleanability and sterility, with integrated CIP/SIP and technical gas filtration systems. A variety of refrigeration options, including liquid nitrogen and eco-friendly air-based cooling (down to -100 °C), deliver performance with sustainability.
Freeze-Drying
IMA offers flexible layout configurations with rear, side-mounted, integrated, or two-storey condenser setups to adapt to any facility footprint and/or process flow. The cooling system skid can be installed on either the upper or lower technical floor, offering even greater flexibility in plant design and space management.
Freeze-Drying
Freeze dryers can be seamlessly integrated into isolator-based lines or other containment solutions and connected to automated loading/unloading systems, ensuring sterility and grade A continuity, avoiding manual intervention and improving overall efficiency.
Freeze-Drying
Completing this offering are IMA Life’s ongoing innovations in lyophilization – including advanced controlled nucleation technology and eco-friendly air refrigeration – ensuring that we remain at the cutting edge of freeze-drying capabilities. Advanced controlled nucleation improves ice structure uniformity and cycle consistency, optimizing drying performance.
In response to ever-changing market demands to reduce human intervention (and hence contamination risk), we have also developed a range of automatic and semi-automatic loading and unloading systems to streamline vial handling.
Innovation continues at IMA Life’s Lab4Life R&D laboratory in Tonawanda (USA), supporting customers with process development, scale-up, and qualification. In parallel, IMA pioneers continuous aseptic spray-freeze-drying, addressing the needs of next-generation biologics and injectable therapies.
Enhance the performance of your LYO and loading/unloading systems, remove obsolescence, extend their lifespan, and achieve environmental and cGMP compliance with a comprehensive set of upgrade services covering machine componentry, refrigeration and vacuum technologies, equipment controls, and all other ancillary systems.
Apply LYO-ReGen to your freeze dryer to ensure compliance with the latest and most stringent regulations. Improve the environmental performance and protection of your equipment and increase productivity through sequential cycling cycles and customization of loading and unloading systems.

• Recipe versioning
• SQL reporting
• Autowithin
• Instruments redundancy
• Control system SCADA
• Control system SCADA & PLC
• Safety device/interlocks
• Non acqueous solvent
• Refrigerant retrofit
• CRF 21 compliance
• Data integrity and cyber security
• Reduce operator intervention
• Reduce equipment downtime
• Fit the reports generated by equipment’s SCADA
• Enhance process robustness and data collection recording
• Enhanced compliance with the most recent requirements for open product handling as per Annex I
• Enhanced compliance with the current environmental and safety regulations
Upgrade your machine with new features that are only available in the “new generation” of freeze dryers. Control and optimize all production phases by connecting to the plant database.

• SQL reporting
• Integration in domain
• Instruments redundancy
• SCADA redundant architecture
• OSI PI – WERUM – MES system
• Continuous Data collection
• Ehnanced uptime
• Batch Report can be generated again at a later time from SQL database,
with indication COPY as water mark
• System under customer IT policy
Be ahead of the curve and ensure full compliance with the new Annex I requirements by upgrading the control system, critical instrumentation, and lyophilizer loading and unloading systems to meet the latest and most stringent environmental control standards and regulatory expectations.

• Instruments redundancy
• Control System upgrades
• Fully automatic Water Intrusion Test for sterilizing filters
• Continuous automatic monitoring and alarming of the sterile boundary of the lyophilization process
• Cycle queuing to enable the fully automatic unattended execution of all service cycles (defrosting, CIP/SIP, Filter integrity test and Unit leak test) to minimize equipment downtime between two consecutive manufacturing cycles while meeting the most recent GMP best practices.
• Unique batch report for freeze drying and services cycles
• Upgrade of the loading/unloading systems (from fully manual to enclosed and ventilated semi- or fully automatic loading/unloading systems) and implement physical barrier technologies around your unsealed containers to support the achievement of Grade A conditions throughout the handling of the partially closed/unsealed containers.
• Enhance your Aseptic process robustness by minimizing the risk of biological
contamination at this crucial step of your aseptic processes
• Automate parts of the handling to minimize equipment/Operator interactions
• Achieve full compliance to Annex I’s requirements and most recent GMP best practices with a reduced capital investment and downtime
The LYO-ReGen package makes it possible to upgrade any machine and eliminate the risk of obsolescence of old components by improving performance or adding features that are only available in “new generation” equipment.

• Mycom
• Cumulus
• Hydraulic power unit & cylinder Control system
• Pneumatic valve manifold from pressure switch to pressure trasmitter
• Vacuum pump (oil or GV to GXS)
• Refrigerant retrofit
• ETV (sporlan to danfoss)
• Reduce unexpected equipment failure
• Improve troubleshooting capability
• Predict downtime due obsolescence of failing items
• Reduce Global warming potential impact
• Customize the stoppering pressure based on vials size
LYO-ReGen reduces energy consumption and limits the impact on the greenhouse effect. If you use non-aqueous solvents, it also helps with liquid separation and waste management.

• Cumulus
• Refrigeration leak detector
• Safetherm HX
• Non acqueous solvent
• Refrigerant retrofit
Reduce environment impact
Immediately detect leakage
Comply with E&S regulation
Prepare for new technology product
– Upgrading existing control systems (PLC and SCADA) to meet the latest requirements and make use of the latest technologies.
– Machine upgrades that eliminate the risk of obsolescence of old components, improve performance, and add features only available in “new generation” equipment.
Improved GMP solutions for existing loading and unloading operations between existing filling and capping equipment and existing freeze dryers.
– Introduction of the latest EM equipment and improved ergonomics and monitoring of RABS access on existing equipment.

The same LYO service products are applicable to the vial loading and unloading systems. However, the following specific features exclusively apply to the vial loading and unloading systems LYO-ReGen package:
Reliability:
• Components (motors, drivers, gearboxes and sensors,
E-cabinet)
• Mechanical disconnect between infeed starwheel and conveyor
• Laser safety scanner
• Laser distance measurement
Performance:
• Mechanical disconnect between infeed starwheel and conveyor
• Laser safety scanner
• Laser distance measurement
Annex 1:
• Semi-Automatic solutions between existing equipment
• Reduced operator activities during loading and unloading of LYO
Optimize Grade A path
• Improved RABS access
• Latest EM devices
Same value and benefits as the freeze dryers. See all benefits listed in the other categories.
The LYO-ReGen package includes a complete update of the control system. The freeze dryer is upgraded to take advantage of the new recipe version management and SQL-based report generation features, as well as all the additional features typical of the “new generation” LYO. Plus, you’ll never again have to worry about potential hardware failures causing a “gap” in automatic cycle reports.
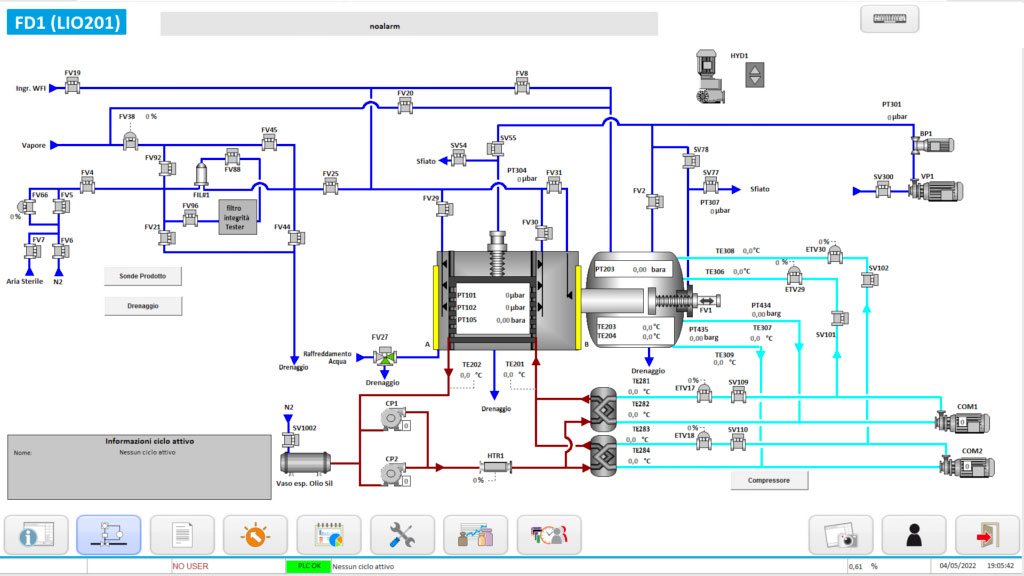
• Virtualization
• SQL reporting
• Integration in domain
• Full control system upgrade
• Redundant architecture
• Multiple freeze dryers
• Stratus server
• ANNEX I compliance
• Batch report can be generated again at a later time from SQL database, with indication COPY as water mark
• Process data for data table and chart(s)
• Alarm filtering for audit trail
• Continuous data collection
• Ehnanced uptime
• Disaster recovery & business continuity
• System under customer IT policy
LYO-ReGen is the upgrade program that allows you to improve the efficiency of your equipment and bring it back to its original performance. It shortens production cycles by automating filter testing and incorporating it into sequential cycles.

• Mycom (from bitzer)
• Cumulus
• Autowithin
• Refrigerant retrofit
• ETV upgrade
• Enhance performance
• Enhance reliability
• Improve equipment up time
• Comply with E&S regulation
Freeze-Drying Solution
Lyophilization, or freeze-drying, is a vital downstream process used across the pharmaceutical and biotech industries to stabilize compounds, extend shelf life, and preserve product efficacy. At the forefront of this field is IMA Life’s Lab4Life, our state-of-the-art lyophilization development laboratory in Tonawanda, USA.
Here, a team of experts partners with customers to accelerate process development, scale-up, and qualification, bridging the gap between research and manufacturing. More than a service lab, Lab4Life is a center for innovation — where new ideas take shape. Ongoing projects include breakthroughs in controlled nucleation and the development of eco-friendly air-based refrigeration systems, both redefining the future of sustainable and high-performance freeze-drying.

Freeze-Drying Solution
From bulk products to vial or tray formats, IMA Life’s industrial freeze dryers are purpose-built to meet the demanding requirements of pharmaceutical, biotech, and diagnostic manufacturing.
Designed for reliability, precision, and aseptic performance, our systems handle a wide spectrum of sensitive products — including vaccines, antibiotics, cytostatics, biologics, hormones, active ingredients, and reagents. Whether for large-scale production or niche applications, every unit is engineered to deliver consistent, high-quality results under the most rigorous standards.

Freeze-Drying

Freeze-Drying

Freeze-Drying Solution
Responding to the rise of personalized medicine, orphan drugs, and high-value small batches, IMA Life offers a comprehensive range of GMP-compliant small-scale freeze dryers designed for agility without compromise.
These compact systems deliver the same levels of quality, sterility, and performance expected in full-scale production, making them ideal for clinical trials, product development, process cycle optimization, and scalable technology transfer to commercial manufacturing.

Freeze-Drying

Freeze-Drying

Freeze-Drying Solution
These systems represent a connection between a typical bench-top freeze dryer used in laboratories and the industrial range of freeze dryers and are designed to ensure the same technical performance and quality standards as a production-scale freeze dryer.
Built to support modern pharmaceutical innovation, they bridge the critical gap between R&D and production with unmatched reliability and flexibility.

Freeze-Drying

Pharmaceutical

Pharmaceutical

Freeze-Drying

Freeze-Drying

Freeze-Drying

Freeze-Drying

Freeze-Drying

Freeze-Drying

Freeze-Drying

Freeze-Drying

Freeze-Drying

6 min
Brands: IMA S.p.A. – LIFE Division Bologna, IMA Life
White paper
18 min
Brands: IMA S.p.A. – LIFE Division Bologna, IMA Life North America Inc.
10 min
Brands: IMA S.p.A. – LIFE Division Bologna
6 min
Brands: IMA Life, IMA S.p.A. – LIFE Division Bologna
Case study
4 min
Brands: IMA Life, IMA S.p.A. – LIFE Division Bologna
4 min
Brands: IMA Life, IMA S.p.A. – LIFE Division Bologna
White paper
3 min
Brands: IMA Life, IMA S.p.A. – LIFE Division Bologna
4 min
Brands: IMA Life, IMA S.p.A. – LIFE Division Bologna
3 min
Brands: IMA Life, IMA S.p.A. – LIFE Division Bologna
7 min
Brands: IMA Life, IMA S.p.A. – LIFE Division Bologna
5 min
Brands: IMA Life, IMA S.p.A. – LIFE Division Bologna
12 min
Brands: IMA Life, IMA S.p.A. – LIFE Division Bologna
9 min
Brands: IMA Life, IMA S.p.A. – LIFE Division Bologna
10 min
Brands: IMA S.p.A. – LIFE Division Bologna
6 min
Brands: IMA Life, IMA S.p.A. – LIFE Division Bologna
10 min
Brands: IMA Life, IMA S.p.A. – LIFE Division Bologna
Case study
11 min
Brands: IMA Life, IMA S.p.A. – LIFE Division Bologna
5 min
Brands: IMA Life, IMA S.p.A. – LIFE Division Bologna
White paper
12 min
Brands: IMA Life, IMA S.p.A. – LIFE Division Bologna
White paper
13 min
Brands: IMA Life, IMA S.p.A. – LIFE Division Bologna
11 min
Brands: IMA Life, IMA S.p.A. – LIFE Division Bologna
10 min
Brands: IMA Life
White paper
2 min
Brands: IMA Life, IMA S.p.A. – LIFE Division Bologna
Case study
8 min
Brands: IMA Life
Need more information about this technology and IMA’s solutions for your industry? Fill in the form and let IMA sales experts reach out to you.