
Exhibition
Step into Cognitive Manufacturing
The IMA Ever‑Evolving System invites you into a new era of manufacturing.
Case study
Reading time 4 min

Lab4Life
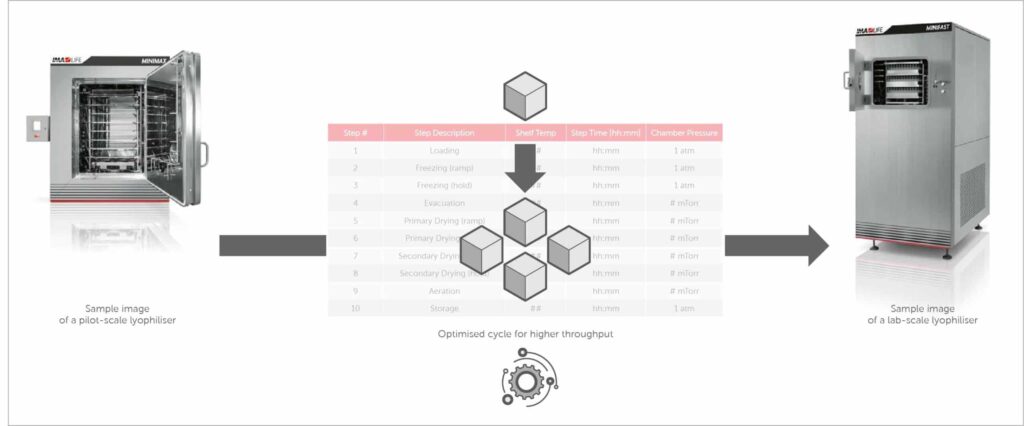
Customer produced a lyophilised hemostatic agent with a legacy cycle using an older lyophiliser. While attempting to increase throughput by changing the fill volume, pressure-control issues arose. Using experiments conducted in IMA’s LYOFAST 3 (2.3 m2), the IMA lab was able to create an optimised cycle providing higher throughput [Stay tuned for future posts to see results from this study!]. It was now necessary to determine if the revised cycle was fit for use in the customer’s newly purchased IMA lyophiliser, a LYOMAX 1 (1 m2) as part of the tech transfer process.
Trays of water were loaded onto each shelf and outfitted with strategically placed thermocouples. Once the water was frozen, the lyophilser was brought down to the lowest possible pressure by the system. Once steady state was achieved, the shelves were brought to a higher temperature, raising the chamber pressure. Once a new steady state was achieved the process was repeated until all desired shelf temperatures were tested.

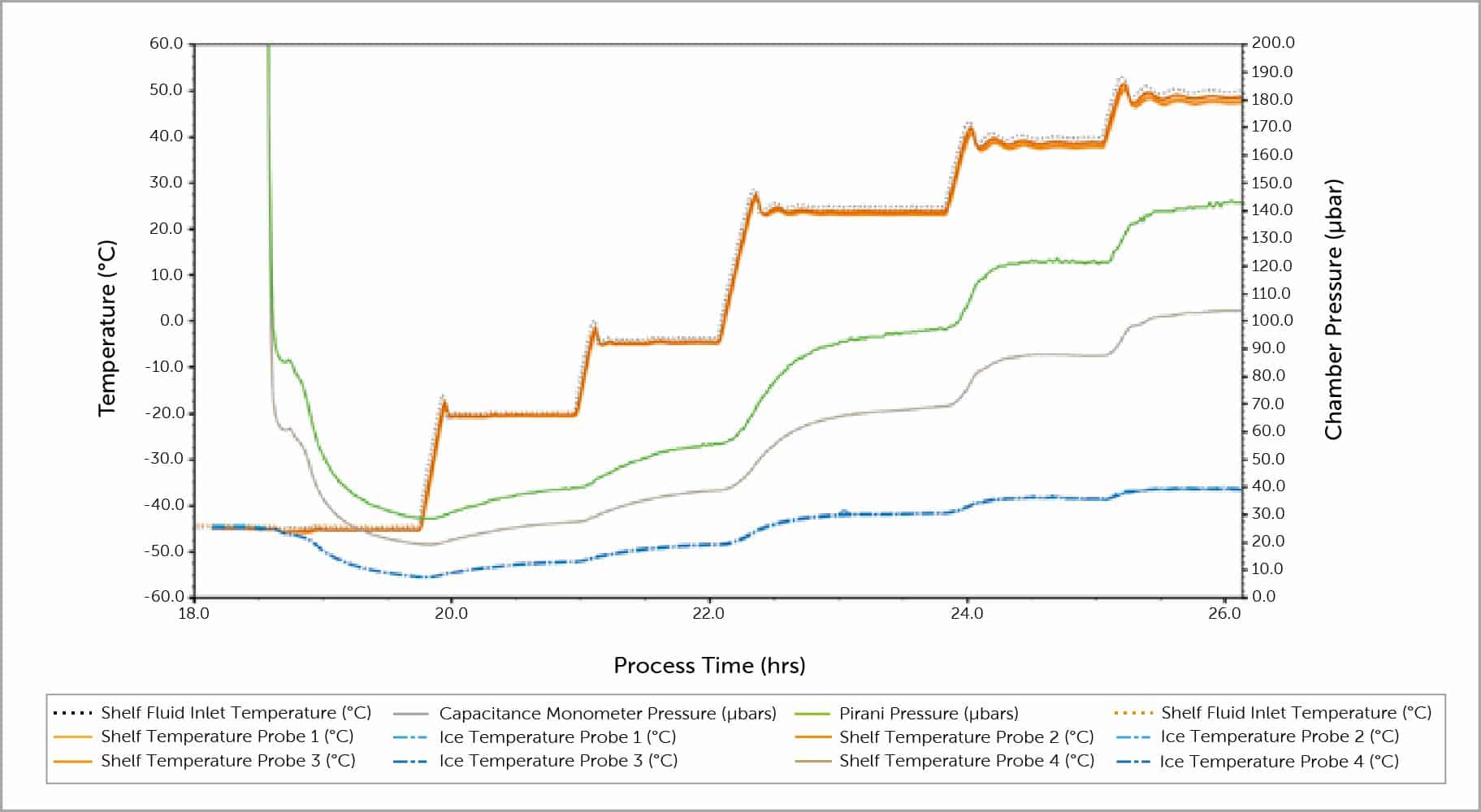
Mass of ice sublimed from each tray along with the corresponding temperature data for the ice at different shelf temperatures during the course of the MCP test was recorded. Using these data and a series of equations characterising mass and heat transfer, an area normalised sublimation rate was calculated for each MCP. The curve generated corresponds with equipment capability of the lyophiliser tested and can be used for constructing a design space.

The parameters of the optimised, higher-throughput cycle developed in the IMA lab lyophiliser were customised via changes in shelf temperatures for use in the Customer’s newly purchased lyophiliser in order to maintain product temperature profiles between the lab and GMP lyophilisers. This ensured that performance intended by the optimised cycle was faithfully replicated.
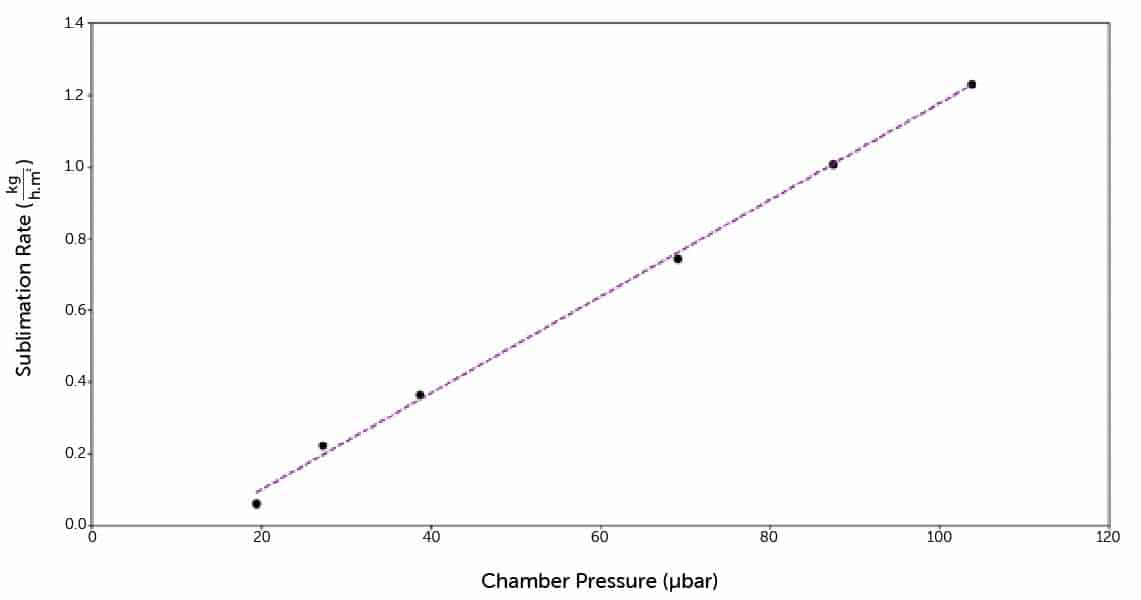
Using collected MCP data and the generated equipment capability curve, it was determined that the Customer’s lyophiliser was capable of running the optimised cycle both in terms of controllable chamber pressure and sublimation rates.
Would you like to learn more about how the optimised, higher-throughput cycle was developed? Stay tuned for future posts!
Lyomax configurable options and custom designs provide the optimal solution for each application.
Lyomax meets the highest possible technical requirements, the strictest standards of the global licensing authorities, and complies with all the current guidelines such as GAMP, cGMP, FDA etc.

The Lab4Life team includes a number of industry experts who provide assistance to customers worldwide with product development, scale up and qualification. To enhance this offering, IMA Life has built a development laboratory at the manufacturing facility in Tonawanda, NY.
