
Exhibition
Step into Cognitive Manufacturing
The IMA Ever‑Evolving System invites you into a new era of manufacturing.
White paper
Reading time 13 min

IMA Active
As the trend for nutritional integrators such as sports supplements and probiotic products grows globally, the IMA Active Division, specialists in solid dose processing within leading pharmaceutical packaging company, IMA Group, has perfected several capsule-filling solutions to cater for the specific characteristics of the nutraceutical sector. Machines and technologies initially developed for pharmaceutical companies, such as high-performance capsule fillers, are proving to bring several benefits to an industry that ranges from local producers to large multinational manufacturers of dietary supplements. This article examines four specific cases, processing of two sports integrators and two probiotic supplements using capsule-filling solutions. IMA Active approaches each case from a single starting point, which is part of the company’s professional policy when dealing with a customer from any industry. The beginning of each case involves the customer in order to get a clear picture of the product and its characteristics, the bottom-line objectives and any of the factors which will impact the decisions made during the stages that go from development to installation. One of the first steps takes place in the IMA Active Competence Center equipped with analytical instruments to ascertain the physical characteristics of the product. This is vital and not only requires certified equipment, but is enriched by the knowledge of the Competence Center team whose experience across the pharmaceutical and nutraceutical industries helps pinpoint the issues that need to be pondered before defining the ultimate solution.
IMA Active has been designing capsule filling solutions since the 1960s. All these years have led to extensive experience and each machine commissioned has added another element of knowledge and perfected the technical aspects. Today’s broad range of capsule fillers is based on a rotary bowl in which the powder height can be set, a dosing unit whose dosator chamber volumes can be easily adjusted and equipped with compression capabilities, and performance within the range reaches an output of 200,000 capsules/hour.
IMA Active’s Competence Center is a hive of activity and staff are constantly assessing products to determine the best equipment and processing parameters to achieve the results desired by the customer, in terms of product quality, processing performance and efficiency. Abiding strictly by the methods and recommendations set out in the United States Pharmacopoeia (USP) and/or the European Pharmacopoeia (Ph. Eur.), and using certified instruments, Competence Center staff examine 3 key aspects regarding powders: density, moisture and particle size. Once these data have been scientifically documented, the Competence Center team is able to use them to ascertain key factors which will impact processing performance and will clarify how to achieve, or even improve on the customer-set targets. These are flowability, measured using the Carr Index, stickiness, fluffiness and the product’s susceptibility to compression.

Equipment used to measure product density:
ERWEKA® SVM 222
This provides valuable information on bulk and tapped density by recording the difference in volume inside the measuring cylinder before and after mechanical tapping.

Equipment used to measure the moisture:
Sartorius® MA160
This determines the amount of volatile matter (water or other liquid) present in the product through the loss of mass when heating it to a fixed temperature.

Equipment used to measure the particle size:
Giuliani® certified sieves
This maps out the particle size distribution and enables the mean value of the particles making up the powder to be established subsequently using defined mathematical calculations.
Let us now take a look at four different case studies which illustrate the results obtained using IMA Active’s capsule filling equipment and technologies.
The first case study regards a widely used supplement, BCAA. BCAA is a mix of three amino acids, in this case without excipients. Laboratory tests to determine bulk and tapped density and thereby detect the flowability of the powder according to the Carr Index confirmed the powder’s classification as poor.
Particle size distribution was measured using certified sieves. Particle size of the amino acid mix is favourable, showing a main particle fraction (39%) between 125 and 180 µm. In this case, the average particle diameter obtained with the sieve method, equivalent to 140 µm, also lies within this same range. Investigation of moisture content using the Loss on Drying (105°C) method also proved the powder to be favourable to processing. It was found to be 0.04% M. The customer set a desired target of 415 mg net weight per capsule, using size 00 capsules, with a tolerance of the individual sample set at <5% of the average gross weight.
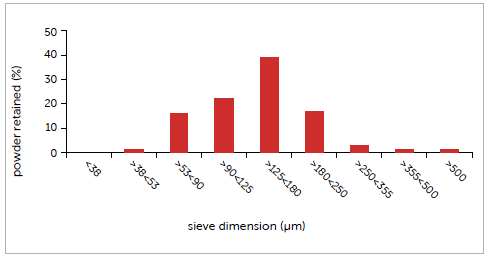
Graph 1: case study #1 particle size distribution.
| Parameters | m.u. | Value |
| Bulk Density | g/ml | 0.382 |
| Tapped Density | g/ml | 0.546 |
| Carr Index | % | 30 (poor flowability) |
Table 1: case study #1 powder characterisation.
| Parameters | m.u. | Value |
| Capsule type/size | mm | Gelatin; size 00 – body length: 20.22 |
| IMA machine | – | ZANASI 40 capsule filler |
| Bowl type | – | Rotary |
| Powder layer height | mm | 45 |
| Dosator material/type | – | Stainless steel with chrome coating |
| Dosing chamber height | mm | 17 |
| Powder compression | mm | 2 inside indicator |
| Mechanical slug release | – | off |
| Achieved output | capsules/hour | 40.000 (max. machine speed) |
Table 2: case study #1 processing parameters.
Despite the fact that the powder’s low bulk density of 0.382 g/ml resulted in an expected capsule content net weight of 347 mg, the target of 415 mg requested by the customer was achieved. Proper conditioning of the powder layer lying 45 mm high in a standard rotary tank was sufficient to manage the potentially negative effects of poor flowability and by applying a modest degree of compression within the dosator, whose chamber height was set at 17 mm in order to keep the dosing level below the rim of the size 00 capsule (h = 20.22 mm). This precaution also avoids soiling the machine during high-speed processing.
IMA uses a capsule filling technology based on alternating motion of the dosing unit. The dosing chamber gathers the powder from a rotary bowl, then rotates and releases the slug into the capsule. This ensures no powder is lost and each capsule is filled with the same quantity throughout the batch. In the case in question,
the customer-set targets were reached without any special need to implement accessories that have been designed to improve performance or overcome the issues generated by nutraceutical products.
The second case study refers to a product with generally good flowability: Acetyl-L-Carnitine containing a 2% excipient (lubricating agent) is a nutritional supplement frequently used by athletes or anyone regularly practising sport.
Table 3 indicates the key data for density and flowability. Particle size distribution was measured using certified sieves, revealing a Gaussian in which 90% of particles are between 53 µm and 500 µm, with an average diameter of 185 µm. The range is broad and such heterogeneous distribution could lead to processing difficulties.
Moisture content was found to be 0.22% M, a favourable factor for keeping processing performance regular over time.
The target established bilaterally after considering the intrinsic characteristics of the powder and other data including capsule size was 793 mg net weight per capsule, using size 00 capsules, with a tolerance of the individual sample set at <5% of the average gross weight.
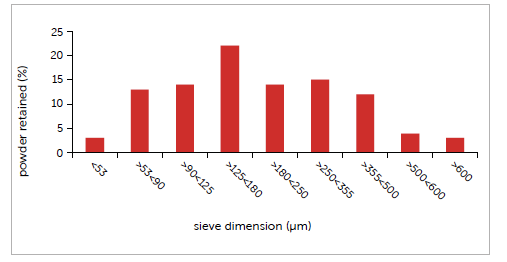
Graph 2: case study #2 particle size distribution.
| Parameters | m.u. | Value |
| Bulk Density | g/ml | 0.694 |
| Tapped Density | g/ml | 0.798 |
| Carr Index | % | 13 (good flowability) |
Table 3: case study #2 powder characterisation.
| Parameters | m.u. | Value |
| Capsule type/size | mm | Gelatin; size 00 – body length: 20.22 |
| IMA machine | – | ZANASI 40E capsule filler |
| Bowl type | – | Rotary |
| Powder layer height | mm | 45 |
| Dosator material/type | – | Stainless steel with chrome coating |
| Dosing chamber height | mm | 21 |
| Powder compression | mm | 2 inside indicator |
| Mechanical slug release | – | off |
| Achieved output | capsules/hour | 40.000 (max. machine speed) |
Table 4: case study #2 processing parameters.
In spite of particle size heterogeneity, the machine performance achieved the expected values: 793 mg net. Formulation composition ensuring good machinability was complemented by the machine’s performance. Considering that a tapped density value of 0.798 g/ml would logically deliver a 726 mg maximum net weight prediction, the processing technology and parameters that were implemented improved the outcome by 9.2% over the maximum based on the powder’s intrinsic characteristics.
Based on collaborative relations with the customer, careful scientific analysis of the powder and the ability of IMA specialists to translate these data into processing parameters, the outcome documented in a full-scale production scenario proved it was possible to exceed the predictable result by almost 10%. This led to increased product quality perception and brought benefits to production in terms of efficiency and consistency.
The third case study deals with a probiotic product whose formulation contains approximately 40% mix of strains (Bifidobacteria and Lactobacilli), approximately 60% prebiotic and a negligible amount of excipients. These microorganisms make a vital contribution to ensuring intestinal microbiota is balanced and healthy, benefiting in turn the immune system and a person’s metabolism.
The dosator tecnology of powder filling in capsule, along with the possibility of gentle pre-compaction of the product with the aspiration bowl, is highly appreciated in the probiotics field: it can properly transfer the product from the bowl to the capsule body with minimum stress or overheating of the probiotic blend. This feature increase dramatically the percentage of live bacteria in the capsule, compared to other capsule filling technologies. A large number of probiotic supplements are administered in size 3 capsules and over 50% of the tests conducted by IMA for this category have been performed on this capsule format.
The Competence Center analyses demonstrated the data in Table 5.
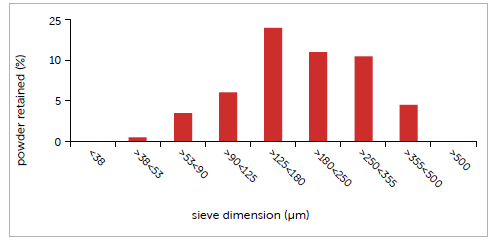
Graph 3: case study #3 particle size distribution.
| Parameters | m.u. | Value |
| Bulk Density | g/ml | 0.493 |
| Tapped Density | g/ml | 0.649 |
| Carr Index | % | 24 (passable flowability) |
Table 5: case study #3 powder characterisation.
| Parameters | m.u. | Value |
| Capsule type/size | mm | Vegetarian; size 3 – body lenght: 13.59 |
| IMA machine | – | PRACTICA 100 capsule filler |
| Bowl type | – | Rotary |
| Powder layer height | mm | 45 |
| Dosator material/type | – | Stainless steel with chrome coating |
| Dosing chamber height | mm | 5 |
| Powder compression | mm | none |
| Mechanical slug release | – | off |
| Achieved output | capsules/hour | 100,000 (max. machine speed) |
Table 6: case study #3 processing parameters.
Particle size of the probiotic powder shows a main particle fraction (28%) between 125 and 180 µm. The results of the particle size distribution analysis carried out using sieves and methodology described in the USP showed a significant heterogeneity of the powder sample: 83% of the particles fall within the following size range: 90 µm to 355 µm and the average particle diameter is 210 µm. The moisture content of the powder, measured with gentle drying
to 105°C was established at 2.31% M. The customer stated they were aiming to achieve size 3 capsules containing 80 mg net weight of product, with a tolerance of the individual sample set at <5% of the average gross weight.
Implementing a powder layer height of 30 mm in the standard rotary bowl without needing to apply compression inside the dosators, the capsule filling machine was able to reach the customer-set targets operating at full processing capacity.
Using standard equipment and without the need to apply compression within the dosator, IMA provided a functional solution to achieve the net target weight of 80 mg. The machine’s base plate is barely soiled during full-scale production as dosing never exceeds the rim of the capsule body.
The fourth case study again treats a product in the probiotic category whose formulation contains a mixture of lyophilised milk enzymes and probiotic strain Saccharomyces Boulardii (approximately 70%), Inulin prebiotic (approximately 30%) and a modest amount of lubricant. Yeasts such as Saccharomyces Boulardii help people stay healthy, defending against external pathogens as well as ensuring correct nutrient absorption for more energy. In order to assess density and flowability, the powder underwent testing on laboratory equipment.
Particle size distribution was measured using certified sieves. The resulting Gaussian shows a main fraction (22%) lying between 125 and 180 µm. Such a wide and heterogeneous distribution, going from 38 to over 355 µm, is an issue that could potentially compromise capsule filling performance. The average particle diameter obtained was found to be 188 µm.
The moisture content of the powder (2.93% M) was determined using the Loss on Drying (105°C) method.
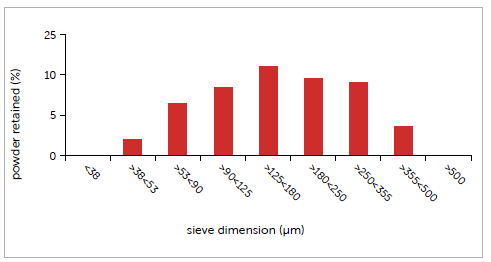
Graph 4: case study #4 particle size distribution.
| Parameters | m.u. | Value |
| Bulk Density | g/ml | 0.601 |
| Tapped Density | g/ml | 0.771 |
| Carr Index | % | 22 (passable flowability) |
Table 7: case study #4 powder characterisation.
| Parameters | m.u. | Value |
| Capsule type/size | mm | Gelatin; size 0 – body lenght: 18.44 |
| IMA machine | – | PRACTICA 100 capsule filler |
| Bowl type | – | Rotary |
| Powder layer height | mm | 47 |
| Dosator material/type | – | Stainless steel with chrome coating |
| Dosing chamber height | mm | 17.95 |
| Powder compression | mm | 3.20 |
| Mechanical slug release | – | off |
| Achieved output | capsules/hour | 90,000 (90% machine speed) |
Table 8: case study #4 processing parameters.
The customer set an ambitious target of 500 mg net weight per capsule, using size 0 capsules, with a tolerance of the individual sample set at <5% of the average gross weight.
Considering the bulk density (0.601 mg/l) of this probiotic powder and its passable flowability (22%), the target set by the customer (500 mg) appeared ambitious; expected net weight of the capsule content was no more than 408 mg. However, the solution provided achieved the desired outcome and enabled production output to reach 90,000 capsules/hour.
Without the need to implement any specific optional features on the ADAPTA 100 capsule filling machine, the project accomplished all targets in terms of performance, efficiency and product quality.
A 47 mm powder layer in the rotary bowl, kept constant with appropriate sensor technology, and a dosing chamber height of just under 18 mm associated with a modest compression of 3.2 mm made it possible to fill the capsules with body height of 18.44 mm thereby limiting powder spillage inside the machine.
With over 6,000 capsule fillers installed worldwide, IMA can claim to have the depth of experience the industry needs to evolve and exploit the latest technologies, as well as the broad knowledge acquired by the IMA Active division in over half a century. Having privileged a technology which implements alternate motion, which can be complemented when necessary by supporting features and technologies the machines adapt intelligently to such a wide variety of nutraceutical products. The machines are also extremely versatile and can be designed to handle different batches with minimal maintenance between one production run and the next. None of this is easily accomplished on machines using the tamping pin technology that require time-consuming cleaning and tool-changing sessions between jobs.
• Aspirating bowl (patented) for hardly compactable powders (light or aerated powders), allowing a constant product bed density and a lower weight standard deviation.
• Infeed with aspirating dosators in case of natural powders herbs, etc.
• Powder slug forced extraction for sticky products.
• Coated dosators for sticky products antibiotics, etc.
• Hardened dosators for abrasive products herbs, etc.
The current range of capsule filling machines is based on the technologies pioneered by IMA and equally so by virtue of the close relationships built with an array of customers over the years. In a market that is experiencing significant growth, responding to increasing demand, where quality of the product is as important to producers as the volumes sold, IMA Active ensures filling precision and consistency. Production flexibility and efficiency also help reduce costs per capsule and enable the industry to keep pace with the demand.
Simple, reliable and effective, Practica is the unbeatable answer to a wide variety of straightforward capsule filling requirements.
