Advanced High-Speed Filling Lines for Liquid and Freeze Dried Products
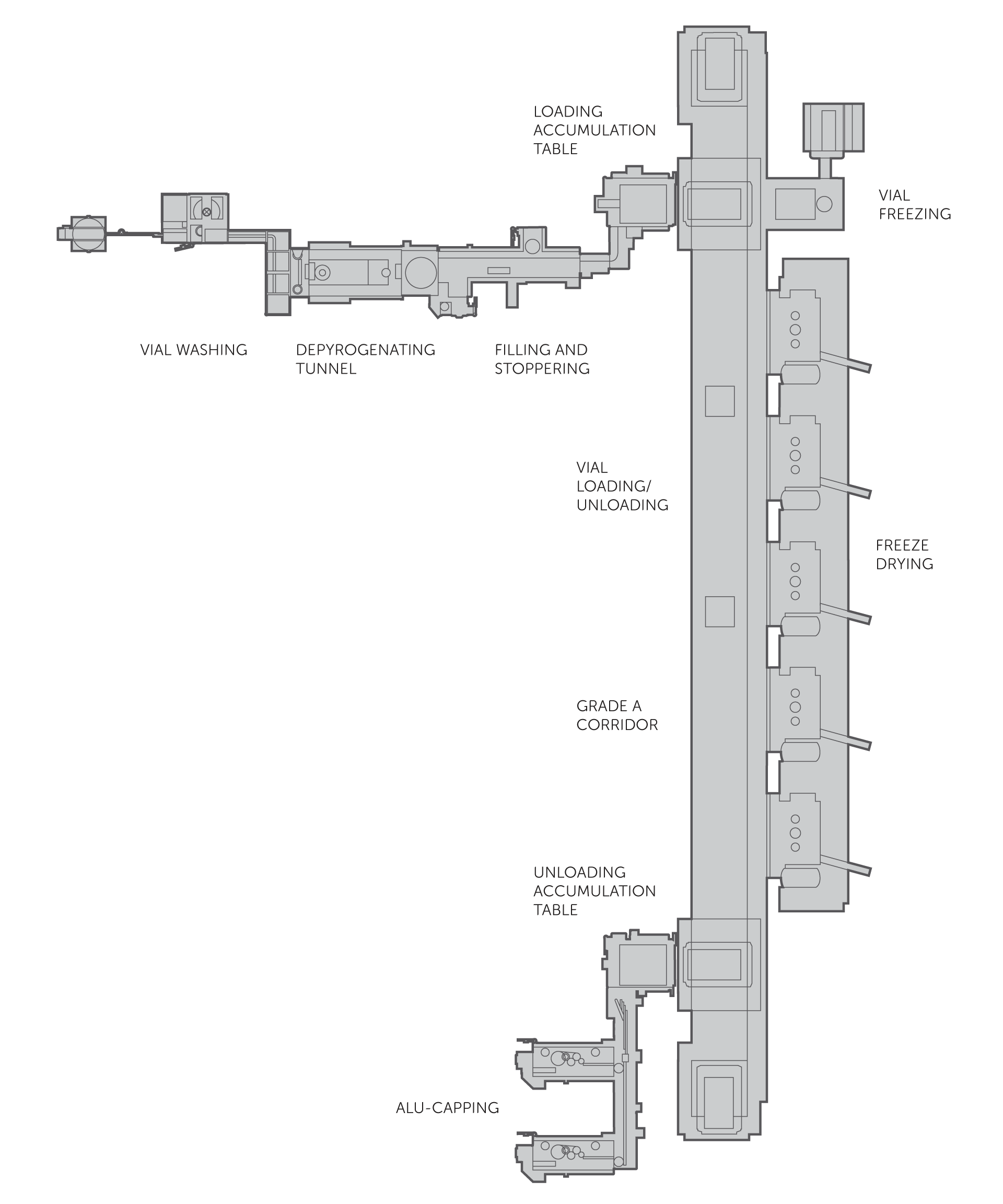
High-speed production plant for vials containing liquid or freeze-dried products, with a throughput of up to 600 vials per minute, starting from washed and depyrogenated bulk containers. The filler uses peristaltic, volumetric or time-pressure dosing technologies, ensuring high flexibility and filling precision with 100% weight control and capping control even at maximum speed. It can be connected to product circuit cleaning and sterilisation (CIP/SIP) systems and nitrogen blanketing solutions with pure nitrogen. Sterility assurance is maximised both by the hygienic design of the filler and by the presence of isolators or RABS optimised to meet the requirements of the new Annex 1, particularly with regard to first air compliance, correct management of sterilised parts in direct and indirect contact with the product and integration with EM. The possible integration of freeze-dryers guarantees extra flexibility, allowing for liquid and freeze-dried product lines to be set up.
Benefits
-
1.
Its suitability for both medium and large batch sizes and large-scale production runs makes this solution ideal for small molecules, vaccines, biological therapies such as monoclonal antibody drugs and antibiotics. The line is divided into modules, each with its own isolator, configurable according to the production process and plant layout. Different isolators can be activated depending on the production goal, such as freeze dryer loading, liquid-only batch production or unloading and capping of freeze-dried products. Each isolator in the system can therefore be configured to manage different production phases and possibly optimise and parallelise production processes across different batches by leveraging the system flexibility.
-
2.
The design of the format-specific parts of the different machines enables format changeover time optimisation, while the installed IPC systems or the refill and restocking strategies minimise the number of rejects and maximise production efficiency.
-
3.
Thanks to the design flexibility of isolators and ventilation systems, high containment applications for high-risk, cytotoxic or biologically active products can be safely handled. VPHP decontamination can be used to ensure microbiological reduction at production start but also the final deactivation of biohazardous products, minimising risks for operators. Products with very stringent environmental control requirements can be managed – including cold (2-8°C) or hot (40-45°C) filled products, products highly sensitive to oxidants and others requiring a pure nitrogen atmosphere.

IMA Life industrial freeze-dryers like Lyomax or Lyofast can be designed, manufactured and qualified to meet any requirement.
Any configuration is possible, whether single floor, multi-level or pass-through. IMA Life freeze dryers embody the requirements of the pharmaceutical industry for faster, safer, cheaper and more ecologically friendly processes:
- The IMA Life patented FusionTM shelves reduce energy consumption typically in heating, cooling and steam sterilization thus achieving a reduction in energy costs and fast temperature response. The FusionTM shelves allow for a 20% reduction in weight leading to reduced energy consumption during freeze drying and sterilization.
- PAT tools reduce overall process time and energy consumption.
- IMA Life is developing innovative eco-friendly refrigeration for freeze dryers, the KryoAir. The KryoAir is a new 100% green refrigeration technology using the ultimate natural refrigerant “air”. The system is based on a series of compression/cooling/expansion steps of air, which can be cooled to a temperature as low as -100° C.



















