IMA for Pharmaceutical
High-Speed Pre-filled Syringe Processing Line
Grade A processing for maximum sterility assurance, high-speed filling up to 36,000 syringes/hr. and the ability to move towards gloveless thanks to latest-generation robotic technologies.
High-Speed Pre-filled Syringe Processing Lines
-
Maximized sterility
6-log decontamination and no-contact robotic handling ensure GMP compliance.
-
High-speed
Processes up to 36,000 syringes/hour with minimal delays
-
Multi-product flexibility
Handles various containers and components
-
Reduced costs
Less downtime, re-qualification, and manual intervention
-
Space-saving
Compact, scalable design fits cleanrooms efficiently
 INJECTA WINNER OF BEST IN SHOW
INJECTA WINNER OF BEST IN SHOW
AT INTERPHEX NY 2023 EXHIBITOR AWARDS

Speed and accurate
processing
A combination of technologies that are revolutionising aseptic processing
Beyond a single choice of machines, IMA takes aseptic processing to a new level with an integrated line made of cutting-edge solutions that interact for maximum performance and security.
Technologies interact,
enhancing each other
An ideal solution for vaccines or monoclonal drugs, both for speed and precision.
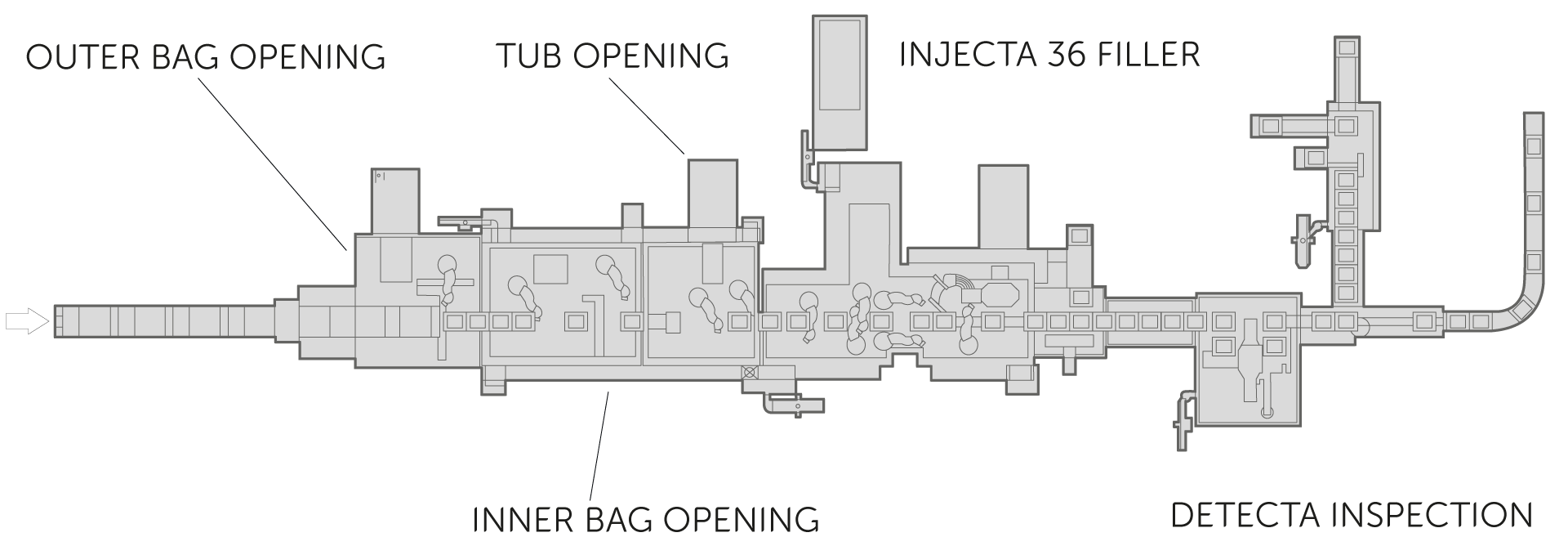
Fully integrated robotic bag/tub/tray-opening is a possible first step to the process before entering the Grade A environment. This can be replaced by the latest decontamination tunnel, NEBULA, which processes batches at high speeds across 3 chambers achieving 6-log decontamination using VPHP. IMA barrier and isolation technologies form an integral part of the aseptic processing line and have been developed to seamlessly accommodate the dynamic processing equipment. In the case of Ready-To-Use syringes, INJECTA 36 will process up to 36,000 nested components in one hour, all operations completed robotically in a sequence implementing No-Touch-Transfer and statistical weight control or 100% IPC. Ensuring accurate dosing at all times, INJECTA 36 also performs automatic, robotic weight cell calibration. Equipped with a plunger presence check using the DETECTA inspection system, the machine enables manual non-conformity rejection downstream during de-nesting, handled by D-NEST. Syringe assembly is than dealt with using MODULA for pre-filled syringes, performing plunger rod insertion, labelling and backstop assembly.
Main features
Designed primarily for large-scale production of pre-filled syringes (PFS) starting from pre-sterilised, Ready-To-Use (RTU) containers, this line combines speed and advanced automation thanks to the use of anthropomorphic robots and innovative solutions for filling and capping. The filler has an in-nest filling mode to handle vials and cartridges in addition to PFS, and is optimised for high-volume pharmaceuticals such as single-dose vaccines for global campaigns or mass-market products for the treatment of diabetes and obesity – such as the latest GLP-1 antagonist medications. Full automation is provided for No-Touch-Transfer (NTT) and opening or continuous decontamination (in Nebula decontamination tunnel) of RTU components. In NTT automated solutions, Ready-To-Use (RTU) containers are fed to the line using automatic unbagging and transfer systems that avoid human contact. In the case of continuous decontamination, RTUs are fed to the line without protective bags, but are exposed to a high concentration of VPHP which reduces their surface charge (6-log reduction) ensuring a grade A rating minimising risks and at the same time avoiding the complexity of robotic manipulation.
Benefits
-
1.
The line offers a complete solution for high-speed production of injectable pharmaceuticals, ensuring maximum patient safety and cutting operating costs. Thanks to flexible use of anthropomorphic robots, the manual procedures required during production can be kept to a minimum and format-specific part requirements can be reduced.
-
2.
Additionally, a compact plant layout is ensured, possibly supplied with an under-isolator solution without HVAC, securing an operational advantage even in small-sized production facilities and designed to also allow integration into existing buildings (brownfields).
-
3.
The use of fully automated, high-speed No-Touch-Transfer (NTT) technologies (up to 5 RTUs/minute) minimises the risk of contamination, increasing product safety, without any impact on plant productivity which can reach up to 36,000 PFS/h.
-
4.
The system flexibility allows different RTU (Ready-To-Use) options to be managed and guarantees high filling precision standards, through an IPC system for weight control that can be operated whenever necessary across all production phases, for statistical or 100% PFS inspection. Injecta 36 can be combined with high-speed inspection systems for stopper presence and position checking inside the syringe or cartridge.
Focus on









