IMA for Pharmaceutical
Flexible Ready-To-Use Component Processing Line
A complete line-up of high-tech solutions that integrate into a perfect sequence of processing and packaging steps for liquids and lyo products.
At every step,
a high-tech solution
Fitting the best technologies together is an additional expertise we leverage.
The key to advanced processing lies in the ability to create an integrated line that incorporates even the most advanced, ground-breaking systems and technologies.
 INJECTA WINNER OF BEST IN SHOW
INJECTA WINNER OF BEST IN SHOW
AT INTERPHEX NY 2023 EXHIBITOR AWARDS
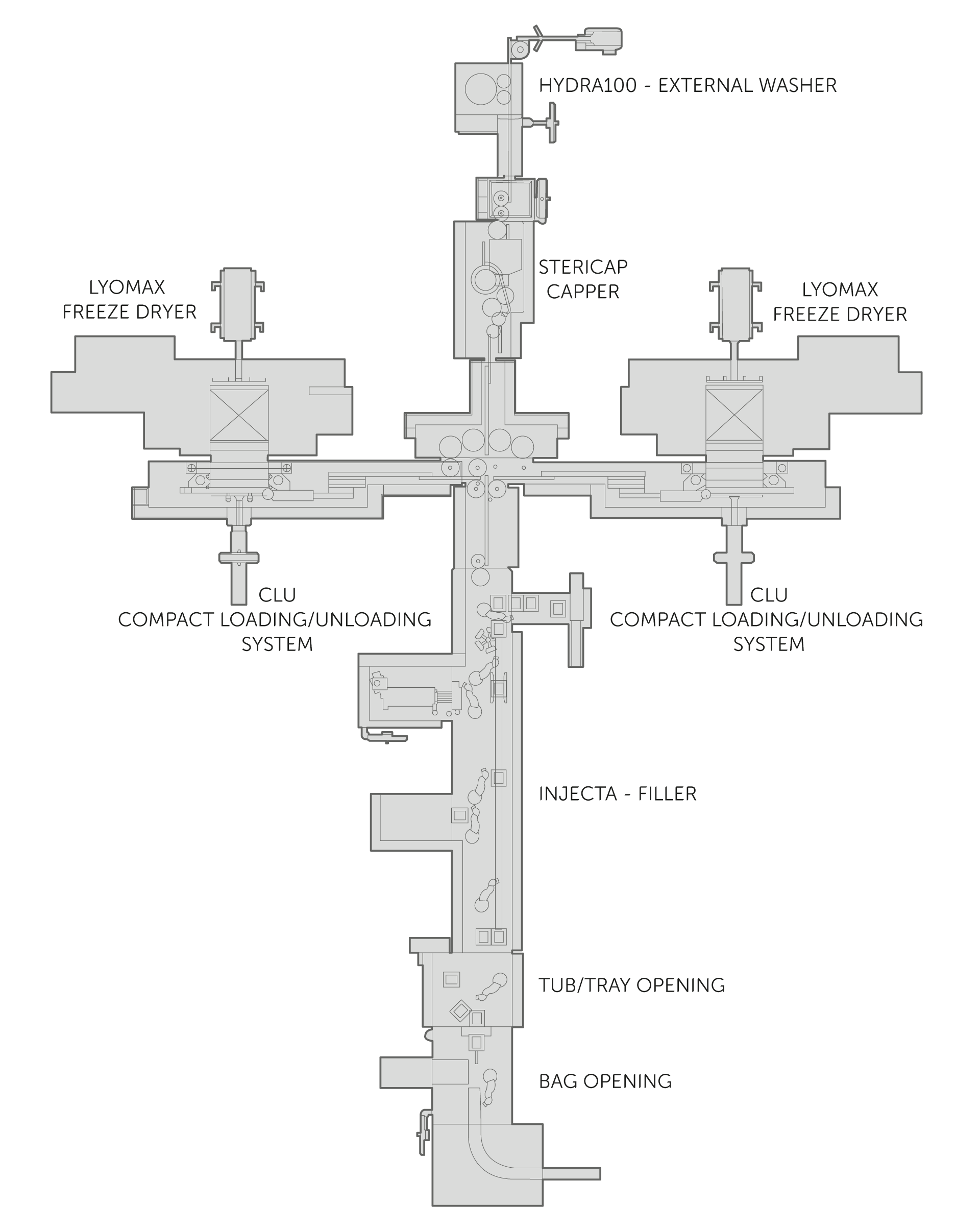
Unique solutions that merge into a future-oriented aseptic processing line.
Starting with upstream preparatory stages as in robotic de-nesting or bag/tub/tray opening, the first section of the line features NEBULA, the high-speed decontamination tunnel achieving 6-log abatement with VPHP. Next up under isolation are the filling operations for multiple formats on INJECTA with its advanced robotics that minimise operator intervention moving aseptic processing closer to a gloveless scenario. Alternatively STERIFILL RS or STERI LIF3 using isolator-barrier systems for sterility assurance and combining filling, stoppering and capping operations. All highly flexible, low to medium-speeds filling of liquids or lyo products, ideal for high-value pharmaceuticals or low volumes with frequent changeovers, and implementing 100% weight control. A complete range of freeze dryers are complemented by the fully integrated loading and unloading systems. LYOMAX, LYOFAST and LYNFINITY can be served by our revolutionary and sustainable KRYOAIR air refrigeration system using ultra low-temperature air as the refrigerant. Vial capping on integrated IMA machines, external washing equipment and tray-loaders fit seamlessly into the line before de-nesting systems complete the aseptic processing line.

Main features
This system is designed for maximum flexibility, allowing various types of containers to be handled, including bottles, syringes and cartridges, starting from pre-sterilised primary containers or RTU (Ready-to-Use) containers. It is particularly suitable for highly potent products (with acceptable concentration requirements up to OEB levels 5 or 6) and biohazardous products (with BSL-2 production site requirements), and can be used for both liquid and freeze-dried product processing. The filler is the Injecta 6 or Injecta 10 robotic solution capable of 100% guaranteed performance at maximum production speed and able to manage different filling technologies such as peristaltic, volumetric or time-pressure dosing. Thanks to its full robotic automation, it can adapt to production runs that require high containment standards. This system is designed to offer maximum flexibility to CDMOs (Contract Development and Manufacturing Organizations), allowing the production of small and medium batches of syringes and vials for both liquid and freeze-dried products. Several innovative technologies are applied, such as No-Touch-Transfer (NTT) and anthropomorphic robots for the handling of RTU containers, eliminating any direct contact with the product and maintaining aseptic process integrity. Thanks to the flexibility of the isolators, the line is designed so as to help monitor containment in terms of filtration, presence of air traps and differential pressure levels adaptable between the various chambers, ensuring that any contaminants do not spread between areas in high containment conditions. The isolators include VPHP-based deactivation methods; an automatic washing system for the isolated chambers and recirculation ducts ensures that there is no residual contamination at batch or campaign end.
Benefits
-
1.
This “multi-mode” solution allows for quick adjustments to production requirements, optimising setup times and helping to safely manage potent, cytotoxic, advanced chemotherapeutic drugs such as the latest Antibody-Drug-Conjugate (ADC).
-
2.
Ideal for customised production and for the management of complex biological medications, this type of integrated system makes it possible to quickly respond to market changes and to production site’s end customer requests. This also results in a significant reduction in waste and a lower risk of cross-contamination between production batches.
Isolated processing line for Ready-To-Use components
Flexibility. Scalability. Modularity. Quality Enhancement. High Containment. Advanced Compliance. Each move targets a specific need. Each move is part of a synchronised strategy. Each move converges into a future-looking concept for aseptic processing, using advanced technologies, latest-generation robotics and patented innovations. Each move leads to Checkmate.













