IMA for Pharmaceutical
Aseptic Ophtalmic And Inhalator Processing Line
Flexible solutions for GMP-compliant processing, including filling, closing and material transfer, and integrating barrier technologies, perfect for sterile ophthalmic substances, nasal sprays and inhalers.
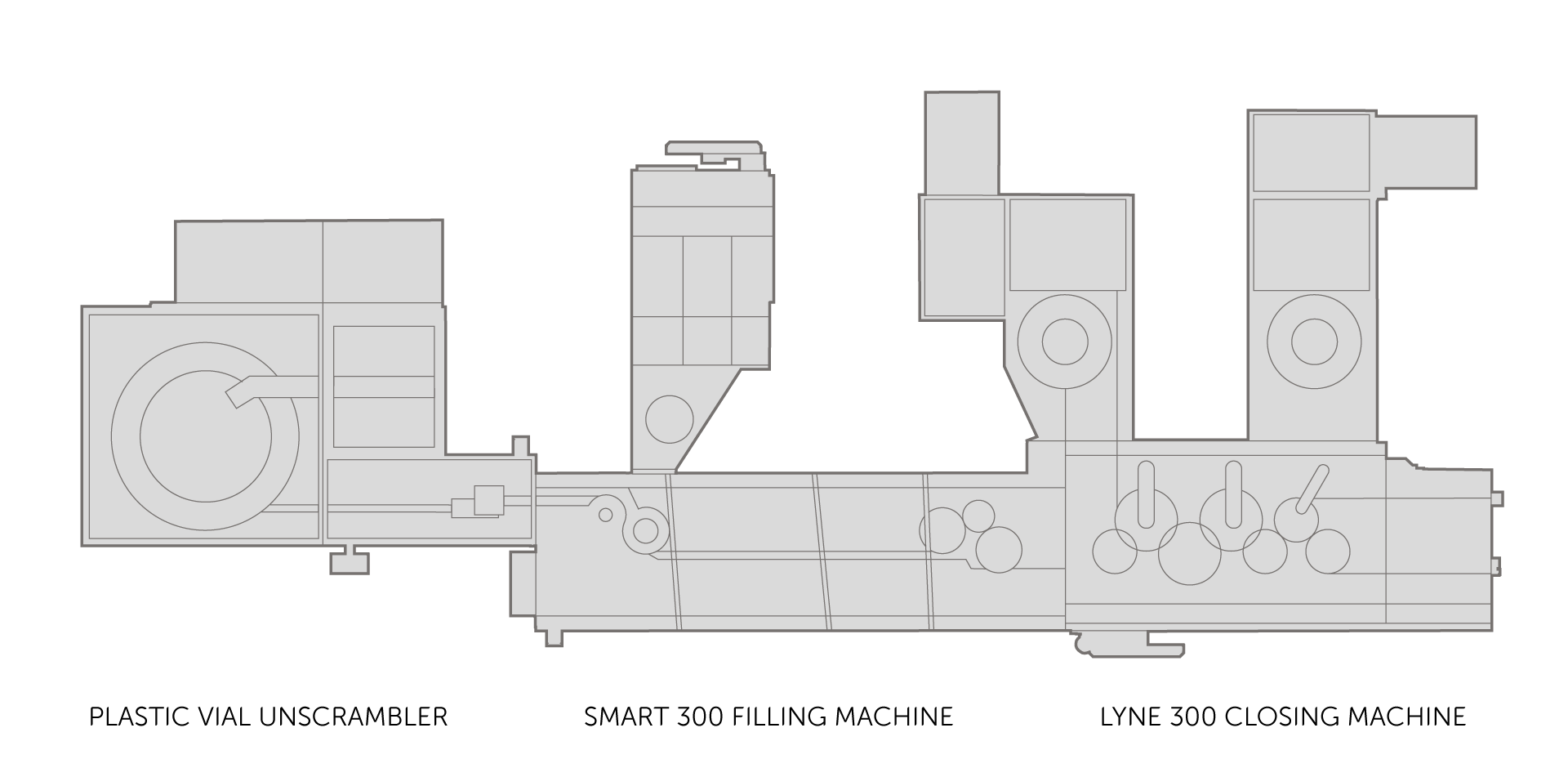
Medium-high speed ophthalmic and inhalators processing lines
Designed for the production of sterile products such as eye drops, inhalers and nasal sprays, this line complies with the new specifications of GMP Annex 1. It is suitable for medium batch sizes, typically between 10,000 and 100,000 units, but also offers enough flexibility to accommodate batch changes and adjust to different sizes. It integrates barrier technologies such as RABS (Restricted Access Barrier Systems) and advanced environmental control systems. This solution guarantees high speed processing of even complex, flexible formats, with multiple components to be assembled. It also implements all the requirements of GMP and the new Annex 1 regarding first air, the management of direct and indirect contact parts requiring steam sterilisation, integration with environmental monitoring and material transfer such as primary containers or filling support material.
Benefits
-
Benefits
1.
It ensures efficient and safe production, meeting the high quality and GMP production requirements of sterile medications for ophthalmic, inhalation and nasal use.
-
Benefits
2
It integrates RABS technology by exactly complying with its stringent guidelines, including the requirement not to open RABS doors during production and to use gloves for all manual operations, ensuring a continuously aseptic environment.
-
Benefits
3.
Using volumetric or peristaltic pumps, the line guarantees precise dosing, adapting to product specifications. Similarly to injectable products, filling of these products is carried out in a controlled environment to maintain the required sterile conditions.
-
Benefits
4.
Thanks to a design that minimises the risk of contamination and to integrated barrier systems, this line meets the top quality requirements of the sensitive products often used for chronic treatments.
-
Benefits
5.
It can be configured for different types of product to be processed on the same system, ranging from ophthalmic products with droppers to nasal sprays equipped with a nebulising pump.






