Case study
Enhancing freeze drying efficiency and product quality through technological and process innovations.
-
Reading time 10 min

-
A study by
IMA Life
Introduction
IMA Life division, leader in the field of aseptic processing and freeze drying solutions, can be considered a true partner to the pharmaceutical and biopharmaceutical industries, offering a wide range of technologically advanced machines.

Freeze-drying, or lyophilization, is a dehydration process in which water is removed from a frozen product by sublimation, the direct conversion of ice into vapor under low pressure. This gentle method enables removal of water at low temperatures, preserving the structural integrity, biological activity, and sterility of sensitive drug substances. Because of this, freeze drying has become the gold standard for stabilizing complex biopharmaceuticals, including vaccines, monoclonal antibodies (mAbs), antibody drug conjugates (ADCs), enzymes, and viral vectors. The essential and increasingly important role of freeze drying in the pharmaceutical industry stems from its ability to extend shelf life and enable global distribution of products that would otherwise require ultra-low-temperature storage. The COVID-19 pandemic highlighted vulnerabilities in the cold-chain infrastructure, prompting regulators and manufacturers to view freeze dried formulations as a cornerstone of supply-chain resilience. By converting unstable liquid formulations into solid, shelf-stable powders, freeze-drying eliminates the need for deep-cold logistics and simplifies transport, while sterility for parenteral administration is maintained through sealed vial closures.

Figure 1: Vials loaded on freeze dryer shelf.
Since 1954, a total of 647 freeze dried drugs have been approved by the U.S.
FDA, reflecting the steady growth and enduring importance of this technology across nearly seven decades of pharmaceutical innovation. Recent regulatory data illustrate this growing importance. Twenty-seven freeze dried drugs were approved by the U.S. FDA in 2023, submitted by 22 pharmaceutical companies.
Excluding discontinued or non-parenteral products, 92% of these approvals were small-molecule drugs and 8% were enzymes. Oncology led all indications (approximately 44%), followed by infectious diseases (approximately 30%), with the remainder spanning diagnostic, rare disease, immune, pulmonary, metabolic, renal, and neurology applications (each about 3.7%)[1].
Freeze-drying is rapidly expanding beyond biologics, impacting nearly every therapeutic area. As the industry advances, innovative technologies and techniques such as design space methodologies and controlled nucleation systems are driving this transformation. These tools improve process reliability, reduce cycle times, and enhance product consistency. Coupled with energy-efficient equipment designs and adaptive control strategies, freeze-drying is evolving from an empirical, resource-intensive method into a precise, data-driven, and sustainable manufacturing platform, positioning it at the forefront of next-generation pharmaceutical production.
Advancements in cycle development: design space method
Advancements in process development and control have enabled a more systematic and predictive approach to freeze-drying cycle design. Historically, cycle optimization relied on empirical methods that required extensive experimentation. With the introduction of Quality by Design (QbD) principles, a structured methodology known as the design space approach has become central to modern cycle development.
A design space defines the multidimensional relationship between key process parameters—such as shelf temperature, chamber pressure, and mass flux—and their combined influence on product behavior during primary drying. Within this framework, scientists can identify safe and efficient operating conditions that maintain product integrity while maximizing throughput, which helps determine optimal settings that prevent issues such as choked flow, product collapse, or incomplete drying.
At IMA Life’s Lab4Life, the construction of a design space for a customer’s formulation typically follows three stages. First, the relationship between chamber pressure and mass flux is established across a range of shelf temperatures to characterize drying kinetics. Second, product isotherms are developed to calculate product temperatures at the vial-bottom under various process conditions to ensure that product temperature remains within acceptable thermal limits.
Third, equipment capability boundaries are determined through computational fluid dynamics (CFD) modeling or empirical testing to define the maximum sustainable mass flux for a given freeze dryer. Integration of this data produces a comprehensive operating map that supports both cycle development and scale-up. Compared to traditional Design of Experiments (DoE) methods, the design space approach reduces the number of required experimental cycles by leveraging modeling and simulation. This accelerates development timelines, enhances process understanding, and aligns with regulatory expectations under QbD. Operating within a scientifically justified design space ensures that drying conditions are optimized not only for efficiency but also for the preservation of critical quality attributes such as potency, structural integrity, and reconstitution behavior.
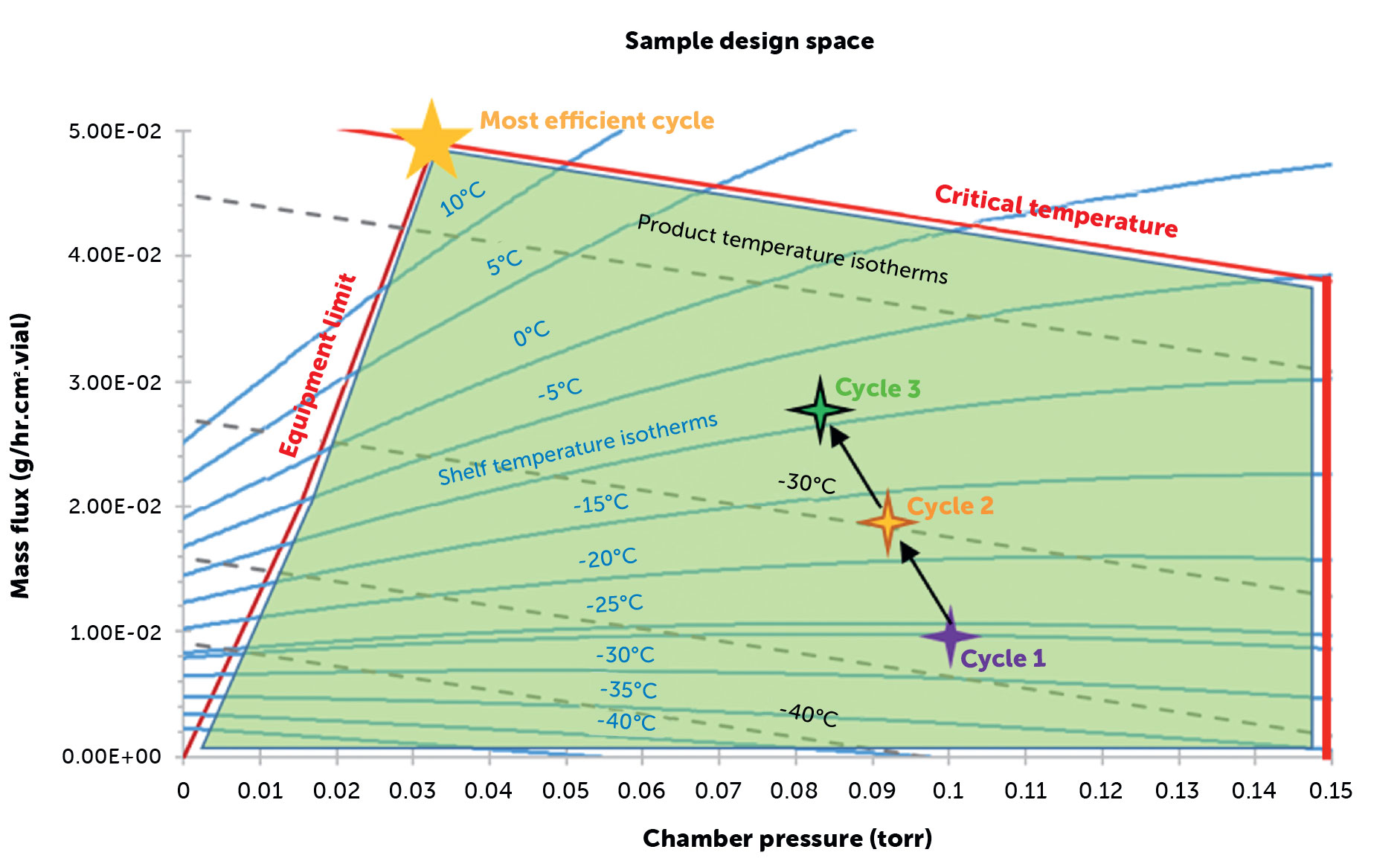
Figure 2: Cycle optimization in design space.
Case study: design space cycle optimization
A practical application of the design space approach was demonstrated by our process development group during the transfer and optimization of a legacy vaccine freeze-drying cycle to a new commercial-scale IMA Life Lyomax 40 freeze dryer (40 m² shelf area). The original cycle exhibited micro-collapse defects that threatened product quality, prompting the implementation of a full QbD-based redevelopment strategy. At the time we initiated this process study, the customer was producing approximately 220 million doses of the vaccine per year and the number of defects was severely impacting manufacturing yield and financial performance. Using baseline process data, a comprehensive design space was constructed to guide targeted modifications across all stages of the freeze-drying cycle. The optimized cycle achieved a 20 % reduction in overall process time while eliminating the previously observed micro-collapse phenomenon. This improvement corresponded to an estimated 44 million additional doses producible within the same timeframe, with significantly reduced risk of batch loss. Testing confirmed that the new freeze dryer could reliably maintain stable control of the process throughout the optimized cycle and within the defined design space, ensuring reproducible product quality and robust process performance. This case study illustrates how the integration of QbD principles and design-space methodology can streamline technology transfer, shorten development timelines, and enhance both scalability and product quality. By transforming empirical cycle adjustments into data-driven decisions, Lab4Life demonstrated the tangible value of modern process science in large-scale vaccine manufacturing.
KRYOSEQ® controlled nucleation technology
In parallel with advances in process understanding and development, technological innovations have markedly enhanced process control, improving both drying efficiency and product quality.
The control and repeatability of freeze-drying cycles are essential for consistently achieving high-quality products. While the primary and secondary drying phases can be well regulated, the absence of control over the ice-nucleation temperature remains a major source of variability. Inconsistent nucleation leads to differences in ice-crystal structure, causing non-uniform drying behavior and variable product stability.
Nucleation is the process by which ice crystals are formed from originating sites called nuclei. In nature, these nuclei are provided by suspended particles or container surface irregularities. In sterile pharmaceutical solutions, however, the absence of such nucleation sites forces the liquid to undergo supercooling before ice crystals can form. The extent of supercooling determines crystal size: liquids that nucleate at lower temperatures generate smaller crystals, whereas nucleation at higher temperatures produces larger, more porous structures. When allowed to nucleate naturally, vials in a batch of product nucleate at different temperatures. Subsequently, these vials dry at different rates and therefore differ in their stability profiles. KRYOSEQ® Nucleation, offered exclusively by IMA Life, is an advanced innovative controlled nucleation technology capable of producing sterile cryogenic ice fog and circulating it within a freeze dryer chamber to ensure reliable and prompt top-down nucleation of the vial-contained pharmaceutical formulations at an optimized nucleation temperature. Due to the ability to induce nucleation at a fixed degree of super-cooling, at a higher product temperature, the resulting ice crystals are larger and more homogeneous which can improve drying rates, reduce vial to vial variance, and often reduce reconstitution times. The KRYOSEQ® process begins by supercooling the liquid formulation to the predetermined nucleation temperature. The chamber is then evacuated to a partial vacuum, after which the controlled injection of pure steam and sterile liquid nitrogen forms the cryogenic ice-fog.
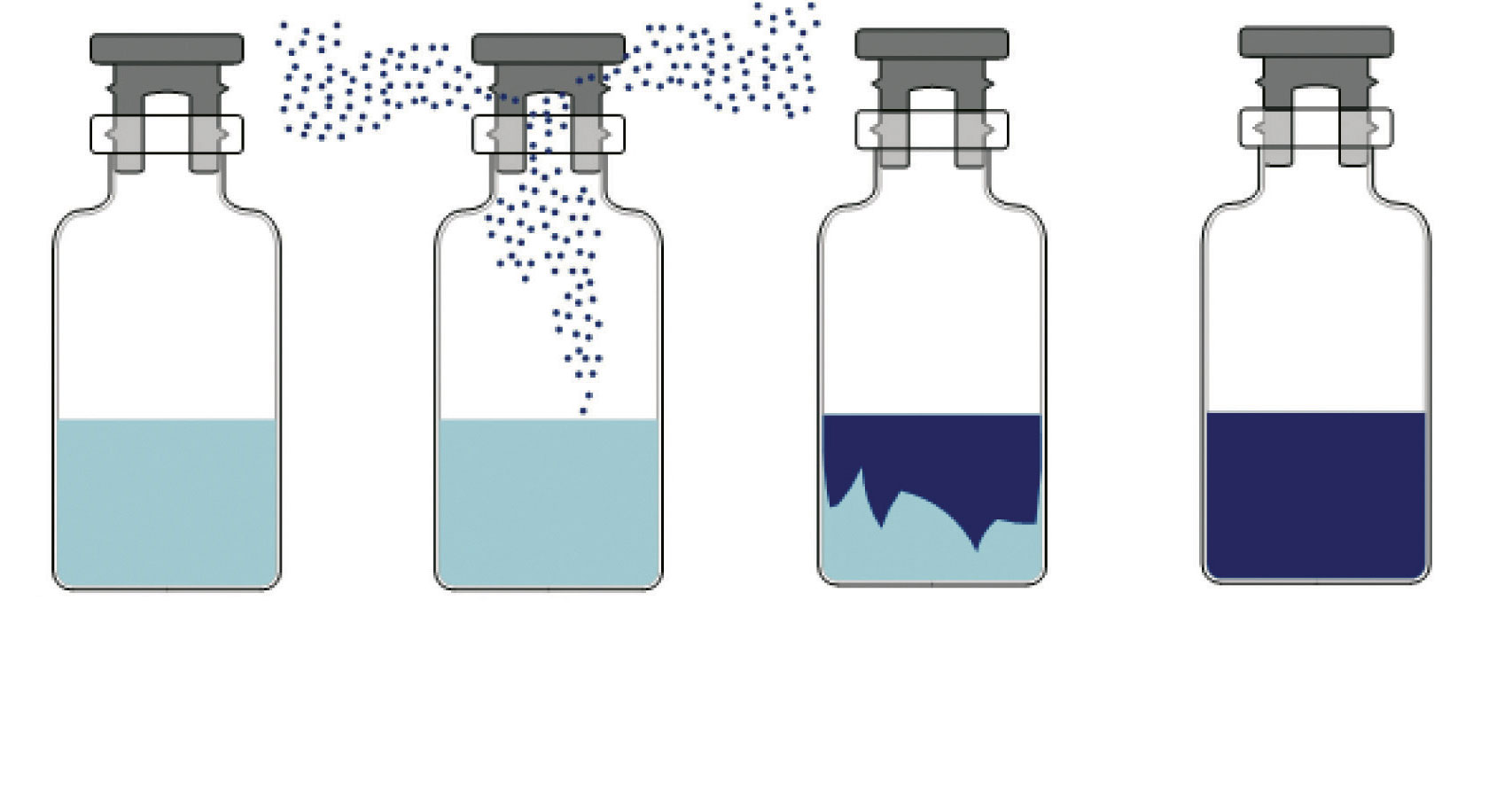
Figure 3: Vial level controlled nucleation process.
This fog permeates the entire chamber and enters each vial, seeding the product surface and initiating synchronous nucleation.
Scalable and retrofittable from laboratory to production-scale systems, KRYOSEQ® provides a robust, aseptically compliant solution for achieving controlled, reproducible freezing, the foundation for consistent product quality and efficient cycle design.
Case study: controlled nucleation in a large-scale freeze dryer
A case study explores performance and process/product impact of the KRYOSEQ® controlled nucleation system in a production scale 56 m² freeze dryer, processing 195,960 vials. The freeze dryer was fully loaded with 3cc vials, each containing a 1.2mL fill, loaded automatically while maintaining a shelf temperature of 5 °C. After loading, shelf temperature was cooled and held at -7 °C for 90 minutes before introducing ice fog. With controlled nucleation, the nucleation temperature range in the monitored vials was -6 to -6.6 °C, where without controlled nucleation, nucleation with significant supercooling was observed. The use of controlled nucleation reduced primary drying time by 19% compared to a cycle with natural nucleation, based on the time required for the last probed vial thermocouple to reach shelf temperature. Testing also showed a 40% improvement in product uniformity, confirming enhanced batch homogeneity and successful scalability of the nucleation system at production scale.
The future of freeze drying
Continuous advancements in modeling and digital technologies are redefining the future of freeze-drying cycle development.
Modern simulation tools are now capable of replicating the complex interactions within a freeze dryer, including heat and mass transfer, vapor flow dynamics, and vial-to-vial variability. Historically, such models were applied only after a cycle to interpret performance; however, the next phase of innovation focuses on real-time monitoring and adaptive control.
Emerging systems aim to establish closed-loop control, where process parameters such as shelf temperature and chamber pressure are automatically adjusted based on in-process adjusted based on in-process measurements and predictive modeling. This approach enables dynamic optimization throughout the cycle, improving efficiency, consistency, and product quality. Ongoing progress in modeling, sensing, and control technologies is paving the way for greater precision, responsiveness and reproducibility in pharmaceutical freeze-drying.
References, bibliography and sitography
[1] Lyohub. (2024). Lyohub Annual Report 2024. Purdue University. Https://engineering. Purdue.edu/
