Injecta Series Aseptic Processing

Machine overview
The future of aseptic processing
Injecta is a state-of-the-art fill-finish machine for RTU components, ensuring superb flexibility for the processing of therapies which require smaller volumes and employ a greater number of different products manufactured in medium-to-low production batches.
Injecta handles pre-sterilised Ready-To-Use containers (syringes, vials or pre-capped cartridges), pre-oriented vials in trays as well as sterilised vials from the depyrogenation tunnel, allowing high process flexibility and adaptability.
Specialised robots perform all handling activities with no glass-to-glass contact and without operator intervention. The result is “less time” spent validating aseptic conditions and superior agility for multi-product manufacturing. The use of advanced robotic-driven manipulations vs. conventional handling systems actually improves product quality and manufacturing efficiency.
Production plant
IMA Life

White paper
IMA Life approach to No-Touch Transfer (NTT) of pre-sterilised Ready-To-Use (RTU) containers.
8 min
Brands: IMA Life, IMA S.p.A. – LIFE Division Bologna
42 min
Brands: IMA Life, IMA S.p.A. – LIFE Division Bologna
Case study
Supporting Merck toward a new age of aseptic fill-finish processing of highly potent drugs.
5 min
Brands: IMA Life, IMA S.p.A. – LIFE Division Bologna
4 min
Brands: IMA Life
6 min
Brands: IMA Life
Machine highlights
-
Robotic handling technologies
Innovative robotics replaces conventional material handling and improves product quality and manufacturing efficiency.
-
100% IPC
Fully robotized 100% check-weighing.
-
Single rejection capabilities
Single rejection of individual non-conformities both from the nest or with denesting.
-
Accuracy and reliability
Precise, accurate and reliable fill-finish process with the highest performance.
-
Containment solutions
Perfectly suited to isolation technology or different containment solutions.
-
Space-efficient
Reduced isolator shell width with positive impact on space ergonomics.
-
Downstream connections
Possible with syringe denesting machine and secondary packaging line.
Superior performance
INJECTA’s ground-breaking innovation is the 100% IPC of all processed products, with single rejection of individual non-conformities both from the nest or with denesting, while ensuring high machine performance.
Technical information
| FEATURES | INJECTA FAMILY | ||
| INJECTA 6 | INJECTA 10 | INJECTA 36 | |
| Automation | Fully robotic automated | Fully robotic automated | Fully robotic automated |
| Footprint | 6,100 x 3,600 x 2,080 [L x W x H] | 7,200 x 4,500 x 2,080 [L x W x H] | 3,400 x 2,050 x 2,080 [L x W x H] |
| In-Process Control | 100% at full speed | 100% at full speed | 100% |
| RTU vial dimensions | Ø 16 – 42.4 mm | Ø 16 – 42.4 mm | Ø 16 – 42.4 mm |
| Vial output | up to 4,800 pcs/h | up to 12,000 pcs/h | up to 18,000 pcs/h |
| RTU syringe dimensions | 0.5 – 20 ml | 0.5 – 50 ml | 0.5 – 50 ml |
| Syringe output | up to 4,000 pcs/h | up to 12,000 pcs/h | up to 36,000 pcs/h |
| RTU cartridge dimensions | 1 – 20 ml | 1 – 20 ml | 1 – 20 ml |
| Cartridge output | up to 3,600 pcs/h | up to 10,800 pcs/h | up to 18,000 pcs/h |
| Suitable for high-potent | Yes | Yes | Yes |
| Loading from depyrogenation tunnel | Yes | Yes | No |
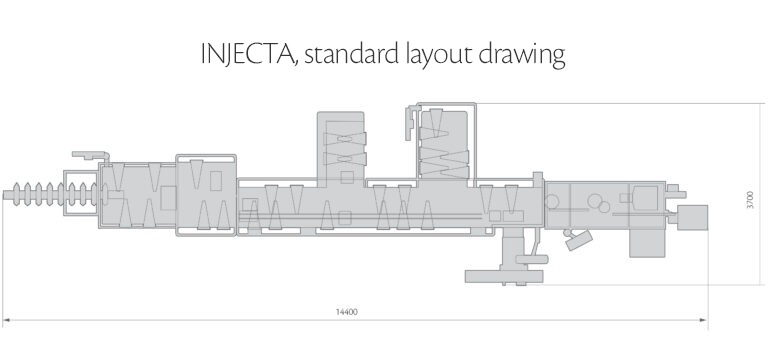
Machine versions
Injecta 36
Injecta 36 is a fine-tuned solution to enhance aseptic fill-finish capabilities and maximise processing line performance. Handling up to 36,000 RTU components per hour, in particular syringes, the system adopts innovative robotic and automation technologies. The use of advanced robotic-driven manipulations versus conventional handling systems actually improves product quality and manufacturing efficiency.

Never miss a move. Follow the winning sequence.
Flexibility. Scalability. Modularity. Quality Enhancement. High Containment. Advanced Compliance. Each move targets a specific need. Each move is part of a synchronised strategy. Each move converges into a future-looking concept for aseptic processing, using advanced technologies, latest-generation robotics and patented innovations.
Each move leads to Checkmate.
A move closer to perfection
INJECTA drives multi-product fill-finish operations into a new realm of flexibility. With its ability to handle so many different product types and formats, and the speed with which processing can switch from one specification to another, INJECTA is the queen of superior versatility.
INJECTA’s strategy of flexibility reflects the need to remain flexible to keep up with changing, patient-centric demands for new drug-delivery systems that can better fit patient lifestyles.
If the primary goal in the current injectable market is to achieve flexible production in a safe, aseptic environment, implementing cost-saving solutions to reduce time-to-market, INJECTA is the winning move. The use of specialised capabilities and the implementation of emerging robotised technologies create a new set of sterilisation, handling and filling challenges, improving product quality and manufacturing efficiency.
The result is “less time” spent validating aseptic conditions and superior agility for multi-product manufacturing: no size part changeover, rise of process capacity, minimal product loss, reduced human intervention, smaller footprint.
#Flexibility is Multi-Product and Multi-format Handling.
INJECTA’s high flexibility for multi-product and multi-format handling enables rapid switches between different drugs and the product’s primary packaging.
- Feeding by Tub/Nest
- Feeding by Tray
- Feeding by Bulk
INJECTA is born to be flexible.
Its filling system can accommodate a wider range of containers like tubs, nests, trays and pre-sterilised RTU components such as vials, syringes and pre-capped cartridges, pre-oriented vials in trays as well as sterilised vials from the depyrogenation tunnel (bulk feeding), allowing high process flexibility and adaptability. No need to change the filling module and minimal/no size changeover is required thanks to INJECTA’s flexible production methods.

#Flexibility is Robotic Revolution.
Whereas conventional attempts to use robots are typically confined to just one step of production, i.e. simply for moving vials from station to station or for removing the cover from a tub of nested containers, INJECTA uses the full potential of its robots.
INJECTA uses the full potential of its robots.
The use of advanced robotically driven manipulations throughout all production operations, from outer/inner bag opening to the stoppering station, allows for a very smooth production process, drastically reducing human interventions and cross-contamination risks; all issues are solved through robot interactions. INJECTA is designed as a whole, exploiting all robotic capabilities in an integrated system. Kawasaki’s seven-axis robot arms are designed for the lowest particle shedding. They are resistant to positive/negative and high pressures and fully compatible for decontamination using H2O2 vapour.
INJECTA’s specialised robots not only provide precise, consistent handling activities, but also offer a high level of flexibility: they are completely digitally controlled with Industry 4.0 capability.
#Flexibility is the Highest Performance in Aseptic Fill-finish Processing.
INJECTA is designed to be flexible in aseptic processing. It can easily combine and perform a fully robotized 100% check-weighing for:
- in-nest configuration
- de-nesting configuration.
INJECTA accurately and efficiently fills and closes RTU components in a nest or with de-nesting.
As a de-nesting solution, INJECTA’s flexible handling units allow for precise removal of components. Individual components are filled, check-weighed, stoppered and then replaced into the nest, in the case of syringes and cartridges, or transferred by a conveyor belt to complete the process.
De-nesting operations are available for high-level quality control, with individual component handling by a robot.
All processed RTU components are individually check-weighed (in the case of de-nesting) or check-weighed all together while in nest, with single rejection of each individual non-conformity, safeguarding the nest.
All the operating units are located in a remote position from the product path with easier access to filling and stoppering areas from both sides of the machine.

A move closer to perfection
Biologics have experienced steady growth over the last 15 years. Similarly, pharma reports on orphan drugs are forecasting that by 2020, six of the ten best-selling global drug therapies will be biologic sterile injectable drugs.
The development of new parenteral targeted medicine for small patient populations and the rising trend for biologics entails big changes for the pharma and biopharma industries.
They will need to adopt flexible and versatile solutions to accommodate smaller batch sizes and, owing to the high value of these drugs, ensure minimal product loss.
Meeting this growing demand for targeted therapies for parenteral delivery, INJECTA is the winning move for a scalable and reliable “medium-to-low production batch” portfolio covering the fill-finish requirements for a wide range of Ready-To-Use disposable components.
INJECTA’s outstanding scalability ensures a unique production platform from the early phases of drug development and clinical trials up to full production scale.
#Scalability is Superior Agility in Aseptic Processing.
Manufacturing targeted biologics requires flexibility and an ability to handle small numbers of high-value batches at an increased frequency. It requires greater process flexibility, versatile equipment, and a more agile approach.
Scalability is what the market is in search of.
As biopharma pipelines grow, there is increasing demand to produce multiple therapies in parallel. All of this means that sterile injectable manufacturing capabilities have to meet the need for improved scalability. This enables flexible, multi-product, small batch production, minimizing product waste and cross-contamination between drugs.

#Scalability is Standardised Design for Container Handling.
INJECTA uses the same supports and transfer devices for all types of RTU components with a unique robotic pincer for material pick&place procedures. This means built-in scalability, high flexibility, simplicity and adaptability in product handling.
Design standardisation helps INJECTA conserve its agility.
Design standardisation helps INJECTA conserve its agility when switching from one container type to another (in nest or with de-nesting), overcoming the complexity and the difficulties of performing aseptic processing with high sterility assurance. INJECTA’s container handling system provides consistency and repeatability, simplifying set up and material transfer without any glass-to-glass contact.

#Scalability is the Same Identical Pattern Repeated for One, Two, Three and up to Ten Filling Pumps.
INJECTA’s robotic multi-format filling system design meets multiple requirements, ranging from small-scale clinical trial batches up to medium-high production levels. Its unique production platform, which implements any number of filling pumps between one and ten, performs the same identical filling, check-weighing and stoppering operations avoiding re-qualification of the production process.
INJECTA, performing the same operations without process re-qualification, enhances your production efficiency.
The filling system implemented on INJECTA relies upon a unique driving system for volumetric and peristaltic pumps and a filling nozzle approved for integration in single-use package forms. Similarly, its 100% check-weighing system can count upon the same HW components for the weighing cells, the same change over parts and the same SW process.
When handling syringes, stoppering operations are performed with the same deep tube and vacuum insertion. In the case of vial stoppering, all operational sequences are identical for both lyo & non-lyo stoppers. When it comes to handling the new integrated rubber & plastic caps, recently introduced on the market, INJECTA handles them in the same way.
The INJECTA family in all configurations ensures unique, standardised sterilisation, filling and stoppering performance, leading to improved product quality and superior manufacturing efficiency.

A move closer to perfection
“Modular facility design” is a hot topic in modern pharma and bio-pharma manufacturing. Such an approach involves designing the process systems as single production units (washing, depyrogenation, handling equipment, filling, stoppering, etc.). These units are generally built and assembled on a parallel timeline with on-site civil and facilities construction. This shortens project time and ensures major advantages such as reduced costs, faster time-to-market and higher quality assembly and fabrication. The modular units are always rearranged with different layouts to add to or form entire plants. To combine intelligent facility layout with innovative processes and technologies, IMA Life not only safeguards set-up modularity (i.e. flexible use of the plant space) but, thanks to our state-of-the-art robotic technologies, also maximises operational modularity.
Modularity is an inherent part of INJECTA’s concept. INJECTA’s integrated “combi” configuration guarantees a complete, contained, integrated production system, with no use of interchangeable production modules to adjust production processes. INJECTA’s design is intrinsically modular and instinctively meets multiple requirements: machine functions can be assembled in different configurations to fit an unlimited number of layout solutions, starting from the handling of primary packaging components.
#Modularity is a High Degree of Automation in Sterile Primary Package Opening.
To preserve the sterile state of the tub/tray, dedicated six-axis robots automatically open the sterile RTU packages and handle all critical aseptic operations. The primary packaging feeding area is usually surrounded by a cRABS area, separating it from the nearby isolated processing area.

Robotic technology ensures great efficiency in handling sterile primary packages also in conventional filling lines.
The tub/tray outer/inner bag opening modules have been designed not only for INJECTA: they can also be integrated with conventional aseptic lines handling RTU vials pre-arranged in trays. This means that small volume RTU vials can also be efficiently fed and processed in traditional aseptic lines, using the same robotic handling system. Forward-looking facilities will consider it an important benefit to best utilize space layout and existing plant conditions to plan for the implementation of a new/future production process. In addition to the given opportunity of scaling existing lines up or down, this would save considerable investment.

#Modularity is Unlimited Smart Solutions for Layout Design Optimisation.
INJECTA allows the optimisation of space design for new parenteral manufacturing and/or for newly designed plants.
Furthermore, in the case of existing plants, thanks to its superior adaptability, INJECTA perfectly fits into an unlimited number of layout configurations with upstream de-pyrogenating tunnel and downstream alu-capping, external washing, up to freeze-drying and complete high potent lines under isolation. In addition, the RTU containers can be accurately and efficiently filled and closed in a nest or with de-nesting. By implementing a modular approach, INJECTA models itself to any kind of production context requirement.
INJECTA can achieve unlimited best-in-class modular layout solutions.
Here are some examples of possible layout configurations:
- Customised solutions to process RTU components (syringes, vials and cartridges) pre-arranged in nest and tub;
- Dedicated solutions to process RTU vials from bulk with de-nesting;
- Dedicated solutions to process RTU vials upside down from trays with de-nesting;
- Dedicated solutions to process RTU vials lyophilised in a nest;
- Dedicated solutions for handling plastic press-fit vial closures (snap caps) for lyo vials.
Modularity is Custom-built Lyo Configurations to Maximise Space Flexibility.
To help them reduce costs emanating from their supply chain, today’s innovative pharma and biotech companies retain flexibility and have the agility to react quickly to changing circumstances within the development, manufacturing and packaging process. The final aim is to combine intelligent facility layout, cutting-edge technologies and innovative processes.
Facilities should allow for flexibility in terms of process development and technical equipment, while maintaining high quality standards. INJECTA helps enhance space flexibility thanks to its own unique intrinsic modular approach.

INJECTA’s intrinsic modularity is fit for lyo process integration.
INJECTA can be easily integrated with an in-line lyophilisation process, where an automated vial loading/unloading system handles products into and out of the freeze dryer. INJECTA’s robotic arm helps place the nested vials into the lyo process ensuring no product losses, no vial falls and no glass breakages. At the end of the lyophilisation process, the pressure applied by the freeze dryer shelves closes the vials. All stoppered vials are then moved to capping operations, relying on conventional standard primary packing components (alu caps).
A few examples of the most frequently required lyo configurations:
- IN-LINE lyo configurations ;
- T-SHAPE lyo configurations;
- All-in-one integrated fill-finish solutions with combined (double lane) lyo process and capping operations.
A move closer to perfection
Pharma companies focus on quality at the beginning of the manufacturing process to ensure against defects and normally employ a quality risk management program to identify critical parameters or conditions that could affect product quality and patient safety, often incorporating operational controls to mitigate or reduce the probability of their occurrence. The principal objective is to determine where particulates are most likely to enter the process, i.e. which are the steps within the end-to-end manufacturing process that are most critical to particle generation and how to control the risk of particle generation.
Managing these risks, though, requires the implementation of a safety program as well as advanced technologies, the use of flexible equipment and proper containment solutions for environmental control.
INJECTA takes quality to a new level, ensuring each step of the process is conducted in the most efficient and quality-oriented way possible.
INJECTA robotic technology offers a means to complete many routine tasks more swiftly, accurately and intelligently, accelerating routine processes and ensuring there are no barriers to agility. INJECTA is beyond doubt an example of how the use of appropriate technology and process automation bring process efficiencies and quality priorities in line.
#Quality Enhancement is Ground-breaking Sterile Outer Bag Handling.
Controlled barriers, containment measures of pressure differentials and protective airflow applied together with advanced robotic handling all help preserve the sterility of the tub (and its components) during outer bag opening procedures. To meet the regulatory requirements, the whole tub-opening unit is completely robotized and under containment. Bag opening is performed in a way that minimises contamination risks, at a point where airflow and pressure difference between two sections ensure the tub remains sterile. After cutting the bag overwrapped around the tub, the primary package is instantly transferred by an internal transfer port to the isolated, processing area.
INJECTA’s robotic technology is a key contribution to preserving tub sterility.
To preserve the sterility of the tub, the bag flaps are opened by suction cups, only when the tub is physically transferred to the isolated area. This innovative pass-through solution helps minimize the exposure of tub and relevant components in less restrictive areas to particles originating from primary packaging materials and/or other routes of particle introduction, improving process quality.

#Quality Enhancement is 100% In-line Process Control with Single Non-conformity Rejection.
INJECTA’s robotic system provides flexibility, ease of use, and sterility assurance enabling a real-time fill-weight check and ensuring the greatest quality result by a single-component rejection system.
INJECTA can easily combine fully robotized 100% check-weighing, while ensuring top-notch machine performance.
All processed RTU components are individually check-weighed (in case of de-nesting) or check-weighed all together while in nest, with single rejection of each individual non-conformity, safeguarding the nest.
INJECTA ensures single rejection both from the nest or with de-nesting.
A patented device used to eliminate the electrostatic charge in the nest is the first step towards accurate weighing, handled by a system designed to minimise interference in weighing accuracy caused by airflow. As a de-nesting solution, INJECTA’s flexible handling units allow for precise removal of the components from the nest. Individual components (1 or 2, or 5-6 at a time) are filled, check-weighed, immediately stoppered, meaning the sterile drug product is subject to much shorter exposure times within the aseptic environment. The design of the 5/6 weigh-cell block also allows for the tolerance required to manage different RTU component formats.

#Quality Enhancement is Maximum Cleansability.
Smaller batches, high potency products such as biologics or conjugates and therapies, involving genes and cells to combat cancer indications, involve the implementation of proper cleansing procedures to avoid or isolate contamination from process areas. INJECTA’s design allows for easy pass-through and cleansing procedures, which are mandatory when handling potent compounds, ensuring the safety and the integrity of the products.
Equipment design, also featuring a massive presence of robots, facilitates protection of open containers thanks to unidirectional airflow.
The overwhelming presence of robots entails the total absence of conventional transfer combs or conveyor belts, facilitating unidirectional airflow (avoiding barriers). Furthermore, INJECTA’s concept design supports proper wash-in-place (WIP) cleansing processes, removing internal surface contaminants from the machine. Surface design facilitates more effective cleaning procedures. Ensuring potential contamination is kept to a strict minimum means greater protection for the product, the equipment itself and the staff operating the equipment or attending to cleaning procedures.
A move closer to perfection
Any drug manufacturing facility handling the next generation of highly active/highly potent compounds targeted for smaller patient populations (HPAPI’s, ADCs) must be equipped with the appropriate methods, controls, and expertise to ensure the safety of its employees and the integrity of its product. Establishing which safety precautions need to be in place to successfully manufacture highly toxic products and avoid any cross contamination within a facility requires a knowledgeable team as well as the design and implementation of an effective safety program to evaluate a compound’s potency and toxicity.
For injectable liquid products, the specific engineering controls to safeguard acceptable daily exposure (ADE) and threshold of toxicological concern (TTC) should be applied at both the facility level and to the production line configuration. The implementation of isolation technology, the use of disposable systems, the installation of flexible and intrinsically modular equipment, the use of air handling systems, coupled with cleaning and bio-decontamination procedures, as well as GMP validation at each stage of manufacturing are imperative to minimize the risk of contamination.
With the goal to maximise operational flexibility, smooth robotic technologies represent not just an evolution of the traditional fill-finish procedures, but a complete re-think of the overall process, offering a new approach to optimize manufacturing. This innovative design approach eliminates the risks and limitations of the conventional actuators and mechanisms connected to the significant amount of format parts necessary to enable this solution to process multiple containers. By the fully robotic approach all format parts have a simple design and are static, relying upon the robot for movement, thus eliminating common sources of particle generation. Therefore, cleaning, sterilization and bio-decontamination of the line between batches is reduced, increasing line performance and enhancing sterility assurance.
At IMA Life, an innovative match has been achieved with the INJECTA robotic filling machine and IMA Life’s isolator engineered containment solution, featuring highly individual customizations, faster format changeover, superior cleanability and ready-to-gloveless, flexible configurations to enable multiple small batch capabilities. Meeting the new HPAPI’s demands in terms of risk mitigation at health-based exposure limits, this is a significant step towards innovative aseptic containment strategies.
# High Containment is having the Perfect Match with IMA Life Isolator System.
INJECTA’s extremely flexible and intrinsically modular design allows switching between production cycles of different pharmaceutical forms (syringes, vials or pre-capped cartridges) enabling the replacement of machine parts more quickly than a standard machine. This challenging solution leads to considerable advantages in isolated lines because, based on the line layouts and on the needs of each individual customer, it allows the isolator to be designed in a more ergonomic way having to interface with a single machine.
IMA Life offers three engineered design configurations—single wall, double wall, and integrated filtration—each tailored to meet specific ergonomic and containment needs in aseptic processing:
SINGLE WALL DESIGN
• Best ergonomics combined with easy glove handling.
• Tailored to specific machine configuration requirements.
• Suitable for high-containment aseptic production.
DOUBLE WALL DESIGN
• Unrivalled chamber visibility.
• No external recirculation channels and flush design.
• Very easy integration inside the cleanroom.
INTEGRATED FILTRATION DESIGN
• Enhanced containment within the aseptic chamber to prevent toxic or hazardous contamination.
• Avoids the presence of air ducts and BIBO filters in the technical compartment.
• Keeps the isolator compact and integrated inside the cleanroom.
INJECTA allows the isolator to be designed in a more ergonomic way having to interface with a single machine.
Thanks to its well-established experience in isolation technology, IMA Life has developed a valuable solution in aseptic containment based on qualified methodologies, control measures of pressure differentials and protective HVAC airflow, meeting EU GMP requirements for product quality, efficacy and patient safety. Furthermore, the containment isolator barrier surrounding the fill-finish processes ensures the processing of multiple compounds in single equipment, a huge advantage when market demands move one way or another, particularly in view of our highly diversified pipeline. Single-use materials such as ready-to-use primary containers, disposable inner/outer bags, etc. are automatically handled and disposed of within the system, with no human intervention. Finally, INJECTA supports a completely automated fill/finish process with the lowest presence of gloves and glove ports, guaranteeing a greater level of sterility.

# High Containment is Design Optimisation.
Thanks to the extreme flexibility of INJECTA, at IMA Life we create the best suited layout configurations according to each customer need and check its ergonomics and design during final review phases. All the activities performed on the line (e.g. assembly of disposable systems, etc.) are tested by a wooden mock-up of the complete filling line and the related isolator, made to 1:1 scale.
INJECTA’s mock-up testing procedure is performed to check INJECTA’s upstream and downstream interactions.
A similar testing procedure is performed to check the interaction between INJECTA and the other units upstream and downstream of the line (e.g. CLU, LUAT etc.), also made of wood and full scale.
Once the mock-up of INJECTA is implemented, the positioning of the utilities inside the isolator (e.g. water, nitrogen, compressed air and steam) is also defined which, thanks to the single block design, do not need to be connected/disconnected during production. The positions of the utilities will therefore be set so as not to affect the ergonomics of the line.
Communication between INJECTA and the isolator requires no changes in the transition from one filling configuration to another, since disconnecting and reconnecting is not necessary in the size changeover phase.
The electronic and electrical connections, just like for the utilities, are defined in the design phase of the machine and tested during the line mock-up phase.

# High Containment is Regular In-place Decontamination.
The unidirectional air flow (UDAF) air in Grade A filling environments suppresses any release of contaminant to a lower level zone, which is contained by the barrier boundary filtration and it is possible to decontaminate in place. The containment barrier is automatically closed for WIP (Washing In Place) cycle which ensures removal of surface contaminants, preventing and neutralizing possible contamination. The improved design of the isolator on a single machine as well as the use of easily cleanable robots capable of moving independently and facilitating the UDAF allows the WIP (Washing In Place) cycle to be optimized. Consequently, considering the worst case filling scenario, it will be adequate to validate a single washing cycle.
INJECTA safeguards a dedicated VPHP bio-decontamination cycle for a single line (INJECTA + isolator).
During the filling line set-up phase, by analysing specific customer production requirements, such as multiple filling processes and production parallelism, it is possible to develop a single decontamination cycle with VPHP. The specific set-points for the decontamination process, once validated, do not require modification or re-testing since it is a dedicated cycle for a single line (INJECTA + isolator).
Developing and validating a single VPHP cycle, for all the possible configurations of INJECTA, results in time saving both for the first validation and for periodic re-validations.

A move closer to perfection
The manufacture of sterile products is subject to special requirements in order to minimize risks of microbiological and particulate contamination. Even if much depends on the skill, training and attitude of the personnel involved, Quality Assurance is particularly important and carefully established and validated methods of preparation and procedure must be followed strictly.
Sterility is a critical quality attribute for all sterile substances, products and containers and must be assured by the use of a suitably designed, validated and controlled manufacturing process.
Sterility is achieved by controlling several factors like the sterilisation procedure, the integrity of the container closure system and, in the case of aseptic processing, the use of satisfactory aseptic techniques.
Reading the Annex 1, “Barrier Technologies” are the most preferred solutions to protect the product from operator and environment, if the drug package cannot be sterilised by terminal sterilisation. Isolator-based technology offers the ability to achieve high sterility levels as it removes the most significant source of microbiological contamination from the aseptic environment by eliminating direct interventions by gowned staff. Typically, a higher Sterility Assurance Level (SAL) is achieved with an isolator system. There are many possible designs of isolators, transfer devices and associated processes: all must comply with the regulatory requirements of FDA/EMA pharmaceutical authorities, so as to provide maximum protection of the grade A environment.
Automation also plays a key role in aseptic fill/finish manufacturing. The existence of a reliable, advanced, process-integrated robotic system makes it feasible to eliminate operator interventions and consequently any contamination potential, while improving efficiency. To be compatible with aseptic manufacturing requirements, robotic systems must be easy to operate, ensure a “near zero particle generating” design and have complete tolerance to in-situ biodecontamination cycles with gaseous hydrogen peroxide. So, we can say that a “towards gloveless isolator solution” is an effect of the presence of highly automated aseptic processing and it means that the production process can run entirely without human manipulations.
In this scenario, INJECTA’s revolutionary isolated robotic technology is a step ahead in the evolution of the fill-finish process, as it allows for superior contamination control. Meeting the Annex 1 requirements, INJECTA’s robotic ability can accomplish multi-product operations by a reliable and reproducible automated process and can also ensure very sophisticated process automation in change parts handling and assembly through RTP devices. The isolator remains the most logical environment in which to place INJECTA’s highly automated intervention-free aseptic fill-finish processes, leading to easier ways to operate and saving on running costs.

#Advanced Compliance is Towards Gloveless Solutions
IMA Life is able to supply complete lines: from the initial washing of bottles, through depyrogenation, filling, capping, etc. … up to and including secondary packaging. All this with or without freeze-drying systems and, in the part where the product is exposed, using an isolator. Isolated solutions are applied to production lines for aseptic and non-toxic products and increase the safety of the final product, ensuring improved aseptic standards and lower operating costs.
All the equipment is produced inside the IMA Life facilities, which is a key factor today. Although we were not among the first to address this market segment, we have come up with a solution that is still unique today. Our isolators have “6 faces” (six walls) in view of the fact that the base of the cube-like structure is also the base of the machine which will then be integrated. This technical detail ensures greater sealing capability, avoiding gaskets between the isolator and the base of the machine, which are present in all other solutions on the market.
Towards a “gloveless” solution implies that the enclosed, fully automated aseptic processing can run completely without human intervention. Therefore, a highly automated machine like INJECTA requires a very minimal presence of gloves around its filling & stoppering “isles” thanks to its reliable, fully robotic system. Some gloves are installed for emergency access or cleaning, but they are not necessary during the production process.
INJECTA is perfectly integrated with IMA Life’s isolator
INJECTA is designed to run with no conveyors, glass-to-glass contact or vibrating bowls which all create risks to the batch.Its multi-axis robotic arms are able to execute the safe, ergonomically uncompromised movement of difficult-to-handle containers offering also repeatability with the flexibility to easily change the process or the container type being filled, so as to efficiently produce high-mix batches. In absence of traditional, mechanical components, decontamination cycles are much shorter too.
“Glovelessness” implies that isolators can run much cleaner than a conventional isolator and be completely closed. All product contact parts are sterilised in place, avoiding need for an operator to directly enter the classified aseptic clean space. All of this drastically reduces the likelihood of a source of microbial contamination. In other words, sophisticated robotic systems operating in an isolator system, can ensure not only outstanding sterility assurance and product safety, but also product outcome reliability in commercial scale production.
INJECTA design is on a pathway leading to fully automated aseptic processing, towards a gloveless system that tends to run without any form of human intervention. Results: cost and time saving, major safety for operators and, thanks to the isolator environment, an increase in product quality and safety.
#Advanced Compliance is Meeting Annex 1 Guidelines
New Annex 1 declaims that the design of the isolator shall take into account all critical factors associated with these technologies, including the air quality of the, the materials and component transfer, the decontamination, disinfection or sterilisation processes and the risk factors associated with the manufacturing operations and materials, and the operations conducted within the critical zone. The best practices for cleaning/decontamination of isolators require that all removable size parts that can be autoclaved (bowls, hoppers, tracks, filling circuit items) are then set in place with a plastic (Tyvek) cover per batch.
By adopting and optimizing robotic technology, at IMA Life we have reduced almost all size parts inside the isolator. Intrinsically an automated robotic line does not need any size parts, such as star wheels and corkscrew formats to guide and move vials. INJECTA’s revolutionary fully automated system can fill multiple container formats and change quickly between vial, syringe and cartridge. Robots can handle every type of RTU container in Nest or Tray in the same way, so there are limited change parts.
As far as the stopper feeding system is concerned, we can count upon an innovative group where the stoppering process is performed using a linear stopper feeding system. Unlike conventional systems, this pioneering group is made of small-sized components ideal for RTP transfer, which is in full compliance with Annex 1 requirements in terms of the sterilisation process. These small-sized parts can be autoclaved and transferred into the isolator via rapid-transfer canisters and ports, without breaking the sterility assurance level.

The use of linear stopper feeding systems reduces particle generation from vibratory bowls
In addition, thanks to its latest breakthrough, INJECTA ensures the fully automated assembly of the stopper feeding unit and of the filling group with no need for human interventions for fine-tuning or pick & place processes. Actually, the autoclaved sterile stoppering/filling parts and components, which are traditionally fed by RTP systems and handled with the use of glove ports, can enter the isolator automatically and be picked & placed by the robotic arms. By this innovative process, clean and sterile parts can be transferred and delivered into the stoppering/filling station inside the barrier system, with leak-tight and risk-free connections, without breaking containment.
So, a towards gloveless highly automated technology provides better aseptic assurance than conventional systems: avoiding the ergonomic constraints imposed by using gloves, the sterile parts and components can be automatically fed and internally assembled with reliability and precision, and all product contact parts can then be sterilised in place.
The use of robotic arms, leads to fewer requirements and easier ways to operate. It increases safety for the operators and the isolator environment, which, in turn, unquestionably increase the quality and safety of the product.
#Advanced Compliance is Real-Time Quality Control Strategies
In the pharmaceutical industry, aseptic processing is monitored—in accordance with regulatory requirements—to prove the environment surrounding a process does not negatively impact product quality and, ultimately, patient safety. Environmental parameters monitored include temperature, relative humidity, differential pressure, and airborne particles. Annex 1, FDA and cGMP’s guidance on Aseptic Processing give special attention to ongoing routine monitoring with regards to the design of systems and setting of action limits alert levels for viable and non-viable environmental and process monitoring. Compliance to these regulatory guidelines, which require continuous monitoring of airborne particle cleanliness, has been a driver for pharmaceutical companies to install facility monitoring systems.
Optical particle counters have historically been the only way of monitoring the cleanliness of the air in real time in pharmaceutical applications. These instruments are installed, based on the principles of quality risk management (QRM) in locations where there is the greatest risk of airborne particle contamination to the process. These instruments could not determine particle viability; they can only count and size particles in real-time.
Recently, the commercialisation of a new technology based on Laser Induced Fluorescence (LIF) has made it possible to look at airborne viable microbial counts in real time. Real-time alarms notify facility operators of alert limits to enable an immediate response to an unwanted event or excursion. Real-time viable particle counters not only offer the potential to monitor these very clean and controlled environments, but can also provide continuous data when integrated into a facility monitoring systems FMS System.
INJECTA is ready to interact with new real-time airborne viable particle counting technologies to quickly react to possible microbial excursions
Approval of this technology will result from discussions between the pharmaceutical companies and the authorising bodies. IMA Life is carefully evaluating applying the technology to the current machines, whereas it will be part of the design criteria for all pipeline projects.
The potential to instantly respond to an airborne microbiological event when it happens is exciting – and beneficial: it improves process understanding and enhances process knowledge. Result is an increase in product quality, a safer product, and risk reduction.

Move into the future of aseptic processing with INJECTA
Building a strategy that addresses major issues and provides a single solution is the goal that has been achieved with INJECTA. Each move evaluates a specific demand facing pharmaceutical and biopharma companies today, answering it with an eye to the future. INJECTA implements advanced technologies, patented devices and relies on the wealth of experience IMA Life has in the field of aseptic processing to say checkmate. One future-looking concept paving the way towards product quality, processing efficiency and production safety.

- INJECTA redefines flexibility and exploits robotic technologies to handle multiple packaging formats.
- Ensuring operational modularity, robots easily adapt to new processing requirements allowing companies to react fast to changing demands. A step further in terms of modularity enables integration with freeze-drying sections or even lines under isolation.
- Enhanced product quality and minimal product losses are obtained thanks to factors such as sterile outer bag handling, airflow and pressure controlled through key design features, patented devices for high-precision weighing and robotized handling under containment
- INJECTA’s unique platform offers unprecedented scalability, adapting to clinical trials or full production.
- High containment capabilities are a must for facilities handling high potent compounds and IMA Life, with its extensive experience in isolation technology, has succeeded in matching its isolator solutions with INJECTA.
- Addressing each of these aspects gains in value for the pharmaceutical company or CMO only when advanced compliance is assured.
- Only a series of coordinated moves leads the Queen to declare Checkmate. Only INJECTA has the power to deliver the outcome required for tomorrow’s world of aseptic processing.
Watch INJECTA video gallery
Experience INJECTA True Gloveless Technology.
Gloveless – Accomplished
Step #1
True gloveless is the ultimate challenge that we are addressing to realise our vision of aseptic processing. It is more about ensuring 100% automation / 0% human intervention than simply removing gloves. Our target is to bring INJECTA as close as possible to the gloveless concept through a step-by-step evolution of technologies, involving our R&D departments and influencing key market players to adopt solutions that will ultimately enable truly gloveless aseptic processing.
So far, we’ve taken the true gloveless concept a step closer to reality, implementing fully automated assembly of the stopper feeding unit and of the filling group. This has been achieved by designing the groups and necessary components so that their assembly is fully managed by robots after entering the initial processing area of Injecta as autoclaved parts inside a stainless steel canister.
Thanks to our pioneering use of robotic technologies, this step of the process has been made truly gloveless.
Gloveless – In the pipeline
Step #2
Moving towards true gloveless processing depends on our ability to exploit existing technologies and to develop new gloveless strategies, as well as on the evolution of complementary technologies and their acceptance by regulatory bodies.
Currently, the continuous viable particle monitoring systems rely on growth-based solutions using settle active plates located inside the processing equipment. We have developed gloveless methods for placing settle active plates in INJECTA’s filling and stoppering section by means of automatic gripper changeover.
This first step has been successfully achieved; however, we believe that the ultimate step toward true gloveless processing will require the implementation of continuous, real-time viable particle monitoring based on BioFluorescent Particle Counter (BFPC) technology. At present, two main challenges remain: the physical size of the spectrometer device and the ability to manage monitoring qualification processes using BFPC in a way that is comparable to traditional active plate methods.
Further progress is therefore required to achieve regulatory acceptance of BFPC technology. IMA Life continues to advance this project and remains keen to collaborate with other SUS suppliers to implement the single-use needle pick-up and assembly solution.
Gloveless – Evolution
Step #3
In the future accomplishing true gloveless processing will depend on our ability to exploit existing technologies and drive innovation towards the ultimate evolution of aseptic processing in an environment in which human intervention is absent. However, achieving such evolution will also hinge on the willingness of other market players to adopt cutting-edge solutions and adapt them to come in line with our vision.
Today, we still count on human intervention to pick up and assemble single-use needles. However, the future has been captured in a solution we designed in-house to make this step an automatic, gloveless operation managed by robots. Designing our own single-use needles kit, we have been able to envisage the answer we expect from the next evolution.
Nonetheless we depend on the manufacturers of single-use kits to join us as we strive to make true gloveless processing a reality for tomorrow’s pharmaceutical industry.
Towards true gloveless
Step #4
What Injecta offers today for pharmaceutical companies seeking maximum versatility in aseptic processing goes beyond the current standards. This is our aim and our challenge, to push back the barriers even further to provide the market with a solution offering both agility and compliance, through advanced robotics to achieve true gloveless processing.
Join the challenge, share the vision and get ready for the next move.
Injecta FAQ
Explore trending topics about Injecta
This is the most demanding changeover in machine configuration. From RTU syringes pre-arranged in TUB & NEST to RTU vials pre-arranged in TRAY, it usually takes no more than 4 hours. Simpler changeovers, when maintaining the same primary package and just changing the internal RTU container, changeover time ranges from 1 to 2 hours.
Is the size changeover controlled by the HMI?
Yes, all the electronically configurable data are stored within each “size recipe”. The mechanical size parts to be replaced are driven by the HMI support.
When changeover is only related to the size of the syringe, vial or cartridge (e.g. from a 1 ml syringe to a 5 ml syringe), how long does it take?
No more than one hour, normally.
We have a patented, in-house solution for the fully automatic opening of the steribag. The cut is made keeping the bag flaps in a closed and fixed position. After cutting, the package is instantly transferred by an internal transfer port to the isolated processing area. To preserve the sterility of the tub, the bag flaps are opened by the suction cups only when they are docked at the gate connecting to the isolated area. The whole tub-opening unit is completely robotised and no operator intervention is required. All operations are protected by unidirectional airflow.
Is this solution applicable in the case of a double steribag (outer + inner bag solution)?
Yes. However, it is imperative that the steribag suppliers ensure an identical geometry and packaging configuration of the bag, thus allowing a completely automatic handling operation.
Can INJECTA handle different steribag materials such as PPT, PPE and so on?
Generally speaking we can handle different materials, yet we believe it fit and wise to arrange internal tests with sample materials provided by the suppliers/customers.
The new Annex 1 guidelines, concerning the sterilisation of the parts in contact with the product, drove our development of an advanced linear plugger/stopper orienting system, avoiding the use of the traditional vibrating bowl. This linear plugger/stopper feeding system is made of small-sized components ideal for RTP transfer by canister with minimal operator intervention. This is in compliance with Annex 1 requirements in terms of the sterilisation process.
In view of the growing demand of the pharmaceutical market to reduce human intervention in sterile handling operations, can this application be defined as a “real gloveless” solution?
We prefer to define our application as a “towards gloveless” solution. At IMA Life we believe that until the authorizing bodies and the pharmaceutical companies commonly agree on the effectiveness of a continuous viable particle monitoring system, to avoid the need of placing microbiological monitoring plates at critical points inside the machine, it is incorrect to use the definition «GLOVELESS MACHINE». Unfortunately, these innovative real-time viable monitoring solutions are still at a development stage.
Does a similar solution already exist for a disposable filling circuit?
Currently we are working closely with all SUS (Single Use Systems) suppliers to complete the filling circuit with pre-assembled nozzles on the supports with quick blocks. For one SUS supplier the solution is already available.
We guarantee a cost-saving solution with a 100% IPC of all processed products with single rejection of individual non-conformities. Furthermore, we fulfil “zero reject” demands at the start and end of each production batch.
Which types of non-conformities can be detected?
We detect individual, non-conforming products caused by wrong dosage and/or plunger/stopper presence or misplacing. On demand, we can also reject the complete tub/tray.
Single rejection can be performed both in the case of a nested solution or in de-nesting.
Injecta Community
Authentication brings you into contact with IMA Life experts and gives you access to reserved material of your own interest.
Our team is ready to welcome the demand for innovation coming from companies and stakeholders, working together to design the future of pharmaceutical manufacturing.
Video interviews
Insight from the experts and all the talent behind the technology.
Publications
Browse our collection of technical papers and learn more about Injecta project developments.
Integrated lines
Pharmaceutical
Flexible Ready-To-Use Component Processing Line

Fill in the form and let IMA sales experts reach out to you.



















